CXCR3 Blockade Inhibits T Cell Migration into the Skin and Prevents Development of Alopecia Areata
- PMID: 27412416
- PMCID: PMC5031416
- DOI: 10.4049/jimmunol.1501798
CXCR3 Blockade Inhibits T Cell Migration into the Skin and Prevents Development of Alopecia Areata
Abstract
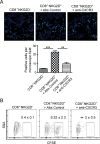
Alopecia areata (AA) is an autoimmune disease of the hair follicle that results in hair loss of varying severity. Recently, we showed that IFN-γ-producing NKG2D(+)CD8(+) T cells actively infiltrate the hair follicle and are responsible for its destruction in C3H/HeJ AA mice. Our transcriptional profiling of human and mouse alopecic skin showed that the IFN pathway is the dominant signaling pathway involved in AA. We showed that IFN-inducible chemokines (CXCL9/10/11) are markedly upregulated in the skin of AA lesions, and further, that the IFN-inducible chemokine receptor, CXCR3, is upregulated on alopecic effector T cells. To demonstrate whether CXCL9/10/11 chemokines were required for development of AA, we treated mice with blocking Abs to CXCR3, which prevented the development of AA in the graft model, inhibiting the accumulation of NKG2D(+)CD8(+) T cells in the skin and cutaneous lymph nodes. These data demonstrate proof of concept that interfering with the Tc1 response in AA via blockade of IFN-inducible chemokines can prevent the onset of AA. CXCR3 blockade could be approached clinically in human AA with either biologic or small-molecule inhibition, the latter being particularly intriguing as a topical therapeutic.
Copyright © 2016 by The American Association of Immunologists, Inc.
Figures



Similar articles
-
CXCL10 produced from hair follicles induces Th1 and Tc1 cell infiltration in the acute phase of alopecia areata followed by sustained Tc1 accumulation in the chronic phase.J Dermatol Sci. 2013 Feb;69(2):140-7. doi: 10.1016/j.jdermsci.2012.12.003. Epub 2012 Dec 26. J Dermatol Sci. 2013. PMID: 23312578
-
Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition.Nat Med. 2014 Sep;20(9):1043-9. doi: 10.1038/nm.3645. Epub 2014 Aug 17. Nat Med. 2014. PMID: 25129481 Free PMC article.
-
Altered T cell subpopulations and serum anti-TYRP2 and tyrosinase antibodies in the acute and chronic phase of alopecia areata in the C3H/HeJ mouse model.J Dermatol Sci. 2021 Oct;104(1):21-29. doi: 10.1016/j.jdermsci.2021.09.001. Epub 2021 Sep 3. J Dermatol Sci. 2021. PMID: 34511266
-
Alopecia areata: Animal models illuminate autoimmune pathogenesis and novel immunotherapeutic strategies.Autoimmun Rev. 2016 Jul;15(7):726-35. doi: 10.1016/j.autrev.2016.03.008. Epub 2016 Mar 10. Autoimmun Rev. 2016. PMID: 26971464 Free PMC article. Review.
-
The potential role of cytokines and T cells in alopecia areata.J Investig Dermatol Symp Proc. 1999 Dec;4(3):235-8. doi: 10.1038/sj.jidsp.5640218. J Investig Dermatol Symp Proc. 1999. PMID: 10674373 Review.
Cited by
-
Functions of NKG2D in CD8+ T cells: an opportunity for immunotherapy.Cell Mol Immunol. 2018 May;15(5):470-479. doi: 10.1038/cmi.2017.161. Epub 2018 Feb 5. Cell Mol Immunol. 2018. PMID: 29400704 Free PMC article. Review.
-
Selective inhibition of JAK3 signaling is sufficient to reverse alopecia areata.JCI Insight. 2021 Apr 8;6(7):e142205. doi: 10.1172/jci.insight.142205. JCI Insight. 2021. PMID: 33830087 Free PMC article.
-
The current state of knowledge of the immune ecosystem in alopecia areata.Autoimmun Rev. 2022 May;21(5):103061. doi: 10.1016/j.autrev.2022.103061. Epub 2022 Feb 10. Autoimmun Rev. 2022. PMID: 35151885 Free PMC article. Review.
-
Immune-mediated alopecias and their mechanobiological aspects.Cells Dev. 2022 Jun;170:203793. doi: 10.1016/j.cdev.2022.203793. Epub 2022 May 29. Cells Dev. 2022. PMID: 35649504 Free PMC article. Review.
-
The CD200-CD200R Axis Promotes Squamous Cell Carcinoma Metastasis via Regulation of Cathepsin K.Cancer Res. 2021 Oct 1;81(19):5021-5032. doi: 10.1158/0008-5472.CAN-20-3251. Epub 2021 Jun 28. Cancer Res. 2021. PMID: 34183355 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

