Tetravalent Dengue Vaccine Reduces Symptomatic and Asymptomatic Dengue Virus Infections in Healthy Children and Adolescents Aged 2-16 Years in Asia and Latin America
- PMID: 27418050
- PMCID: PMC5021228
- DOI: 10.1093/infdis/jiw297
Tetravalent Dengue Vaccine Reduces Symptomatic and Asymptomatic Dengue Virus Infections in Healthy Children and Adolescents Aged 2-16 Years in Asia and Latin America
Abstract
Background: Asymptomatic dengue virus-infected individuals are thought to play a major role in dengue virus transmission. The efficacy of the recently approved quadrivalent CYD-TDV dengue vaccine against asymptomatic dengue virus infection has not been previously assessed.
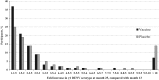
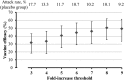
Methods: We pooled data for 3 736 individuals who received either CYD-TDV or placebo at 0, 6, and 12 months in the immunogenicity subsets of 2 phase 3 trials (clinical trials registration NCT01373281 and NCT01374516). We defined a seroconversion algorithm (ie, a ≥4-fold increase in the neutralizing antibody titer and a titer of ≥40 from month 13 to month 25) as a surrogate marker of asymptomatic infection in the vaccine and placebo groups.
Results: The algorithm detected seroconversion in 94% of individuals with a diagnosis of virologically confirmed dengue between months 13 and 25, validating its discriminatory power. Among those without virologically confirmed dengue (n = 3 669), 219 of 2 485 in the vaccine group and 157 of 1 184 in the placebo group seroconverted between months 13 and 25, giving a vaccine efficacy of 33.5% (95% confidence interval [CI], 17.9%-46.1%) against asymptomatic infection. Vaccine efficacy was marginally higher in subjects aged 9-16 years (38.6%; 95% CI, 22.1%-51.5%). The annual incidence of asymptomatic dengue virus infection in this age group was 14.8%, which was 4.4 times higher than the incidence for symptomatic dengue (3.4%).
Conclusions: The observed vaccine efficacy against asymptomatic dengue virus infections is expected to translate into reduced dengue virus transmission if sufficient individuals are vaccinated in dengue-endemic areas.
Keywords: Asia; Latin America; adolescents; asymptomatic dengue virus infection; children; dengue vaccine; symptomatic dengue virus infection.
© The Author 2016. Published by Oxford University Press for the Infectious Diseases Society of America.
Figures

References
-
- Guzman MG, Harris E. Dengue. Lancet 2015; 385:453–65. - PubMed
-
- Capeding MR, Tran NH, Hadinegoro SR et al. Clinical efficacy and safety of a novel tetravalent dengue vaccine in healthy children in Asia: a phase 3, randomised, observer-masked, placebo-controlled trial. Lancet 2014; 384:1358–65. - PubMed
-
- Hadinegoro SR, Arredondo-Garcia JL, Capeding MR et al. Efficacy and long-term safety of a dengue vaccine in regions of endemic disease. N Engl J Med 2015; 373:1195–206. - PubMed
-
- Villar L, Dayan GH, Arredondo-Garcia JL et al. Efficacy of a tetravalent dengue vaccine in children in Latin America. N Engl J Med 2015; 372:113–23. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

