Elevation of iron storage in humans attenuates the pulmonary vascular response to hypoxia
- PMID: 27418684
- PMCID: PMC5007321
- DOI: 10.1152/japplphysiol.00032.2016
Elevation of iron storage in humans attenuates the pulmonary vascular response to hypoxia
Abstract
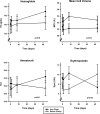
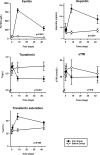
Sustained hypoxia over several hours induces a progressive rise in pulmonary artery systolic pressure (PASP). Administration of intravenous iron immediately prior to the hypoxia exposure abrogates this effect, suggesting that manipulation of iron stores may modify hypoxia-induced pulmonary hypertension. Iron (ferric carboxymaltose) administered intravenously has a plasma half-life of 7-12 h. Thus any therapeutic use of intravenous iron would require its effect on PASP to persist long after the iron-sugar complex has been cleared from the blood. To examine this, we studied PASP during sustained (6 h) hypoxia on 4 separate days (days 0, 1, 8, and 43) in 22 participants. On day 0, the rise in PASP with hypoxia was well matched between the iron and saline groups. On day 1, each participant received either 1 g of ferric carboxymaltose or saline in a double-blind manner. After administration of intravenous iron, the rise in PASP with hypoxia was attenuated by ∼50%, and this response remained suppressed on both days 8 and 43 (P < 0.001). Following administration of intravenous iron, values for ferritin concentration, transferrin saturation, and hepcidin concentration rose significantly (P < 0.001, P < 0.005, and P < 0.001, respectively), and values for transferrin concentration fell significantly (P < 0.001). These changes remained significant at day 43 We conclude that the attenuation of the pulmonary vascular response to hypoxia by elevation of iron stores persists long after the artificial iron-sugar complex has been eliminated from the blood. The persistence of this effect suggests that intravenous iron may be of benefit in some forms of pulmonary hypertension.
Keywords: hypoxia inducible factor; pulmonary circulation; pulmonary hypertension.
Copyright © 2016 the American Physiological Society.
Figures



References
-
- Balanos GM, Dorrington KL, Robbins PA. Desferrioxamine elevates pulmonary vascular resistance in humans: potential for involvement of HIF-1. J Appl Physiol 92: 2501–2507, 2002. - PubMed
-
- Bishop T, Talbot NP, Turner PJ, Nicholls LG, Pascual A, Hodson EJ, Douglas G, Fielding JW, Smith TG, Demetriades M, Schofield CJ, Robbins PA, Pugh CW, Buckler KJ, Ratcliffe PJ. Carotid body hyperplasia and enhanced ventilatory responses to hypoxia in mice with heterozygous deficiency of PHD2. J Physiol 591: 3565–3577, 2013. - PMC - PubMed
-
- Brusselmans K, Compernolle V, Tjwa M, Wiesener MS, Maxwell PH, Collen D, Carmeliet P. Heterozygous deficiency of hypoxia-inducible factor-2alpha protects mice against pulmonary hypertension and right ventricular dysfunction during prolonged hypoxia. J Clin Invest 111: 1519–1527, 2003. - PMC - PubMed
-
- Formenti F, Beer PA, Croft QP, Dorrington KL, Gale DP, Lappin TR, Lucas GS, Maher ER, Maxwell PH, McMullin MF, O'Connor DF, Percy MJ, Pugh CW, Ratcliffe PJ, Smith TG, Talbot NP, Robbins PA. Cardiopulmonary function in two human disorders of the hypoxia-inducible factor (HIF) pathway: von Hippel-Lindau disease and HIF-2alpha gain-of-function mutation. FASEB J 25: 2001–2011, 2011. - PMC - PubMed
-
- Geisser P, Banke-Bochita J. Pharmacokinetics, safety and tolerability of intravenous ferric carboxymaltose: a dose-escalation study in volunteers with mild iron-deficiency anaemia. Arzneimittelforschung 60: 362–372, 2010. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

