Antibody Responses After Analytic Treatment Interruption in Human Immunodeficiency Virus-1-Infected Individuals on Early Initiated Antiretroviral Therapy
- PMID: 27419172
- PMCID: PMC4943535
- DOI: 10.1093/ofid/ofw100
Antibody Responses After Analytic Treatment Interruption in Human Immunodeficiency Virus-1-Infected Individuals on Early Initiated Antiretroviral Therapy
Abstract
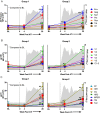
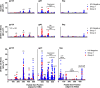
The examination of antibody responses in human immunodeficiency virus (HIV)-1-infected individuals in the setting of antiretroviral treatment (ART) interruption can provide insight into the evolution of antibody responses during viral rebound. In this study, we assessed antibody responses in 20 subjects in AIDS Clinical Trials Group A5187, wherein subjects were treated with antiretroviral therapy during acute/early HIV-1 infection, underwent analytic treatment interruption, and subsequently demonstrated viral rebound. Our data suggest that early initiation of ART arrests the maturation of HIV-1-specific antibody responses, preventing epitope diversification of antibody binding and the development of functional neutralizing capacity. Antibody responses do not appear permanently blunted, however, because viral rebound triggered the resumption of antibody maturation in our study. We also found that antibody responses measured by these assays did not predict imminent viral rebound. These data have important implications for the HIV-1 vaccine and eradication fields.
Keywords: HIV eradication; HIV vaccine; antibody; microarray; treatment interruption.
Figures



References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

