An appraisal of drug development timelines in the Era of precision oncology
- PMID: 27419632
- PMCID: PMC5288167
- DOI: 10.18632/oncotarget.10588
An appraisal of drug development timelines in the Era of precision oncology
Abstract
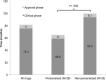
The effects of incorporating a biomarker-based (personalized or precision) selection strategy on drug development timelines for new oncology drugs merit investigation. Here we accessed documents from the Food and Drug Administration (FDA) database for anticancer agents approved between 09/1998 and 07/2014 to compare drugs developed with and without a personalized strategy. Sixty-three drugs were included (28 [44%] personalized and 35 [56%] non-personalized). No differences in access to FDA-expedited programs were observed between personalized and non-personalized drugs. A personalized approach for drug development was associated with faster clinical development (Investigational New Drug [IND] to New Drug Application [NDA] submission; median = 58.8 months [95% CI 53.8-81.8] vs. 93.5 months [95% CI 73.9-112.9], P =.001), but a similar approval time (NDA submission to approval; median=6.0 months [95% CI 5.5-8.4] vs. 6.1 months [95% CI 5.9-8.3], P = .756) compared to a non-personalized strategy. In the multivariate model, class of drug stratified by personalized status (targeted personalized vs. targeted non-personalized vs. cytotoxic) was the only independent factor associated with faster total time of clinical drug development (clinical plus approval phase, median = 64.6 vs 87.1 vs. 112.7 months [cytotoxic], P = .038). Response rates (RR) in early trials were positively correlated with RR in registration trials (r = 0.63, P = <.001), and inversely associated with total time of drug development (r = -0.29, P = .049). In conclusion, targeted agents were developed faster than cytotoxic agents. Shorter times to approval were associated, in multivariate analysis, with a biomarker-based clinical development strategy.
Keywords: FDA; biomarkers; drug development; pharmacoeconomics; precision medicine.
Conflict of interest statement
Razelle Kurzrock has research funds from Sequenom, Foundation Medicine, Guardant, Pfizer, Merck Serono and Genentech, is a consultant to Actuate Therapeutics and Sequenom, and has an ownership interest in Novena Inc. and Curematch Inc.
Figures


References
-
- Hutchinson KE, Lipson D, Stephens PJ, Otto G, Lehmann BD, Lyle PL, Vnencak-Jones CL, Ross JS, Pietenpol JA, Sosman JA, Puzanov I, Miller VA, Pao W. BRAF fusions define a distinct molecular subset of melanomas with potential sensitivity to MEK inhibition. Clin Cancer Res. 2013;19:6696–6702. - PMC - PubMed
-
- Krampitz GW, Norton JA. RET gene mutations (genotype and phenotype) of multiple endocrine neoplasia type 2 and familial medullary thyroid carcinoma. Cancer. 2014;120:1920–1931. - PubMed
-
- Food and Drug Administration and US Department of Health and Human Services. Critical Path Report. 2006 Mar; http://www.fda.gov/downloads/ScienceResearch/SpecialTopics/CriticalPathI...
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

