Cardiac Chemical Exchange Saturation Transfer MR Imaging Tracking of Cell Survival or Rejection in Mouse Models of Cell Therapy
- PMID: 27420900
- PMCID: PMC5207119
- DOI: 10.1148/radiol.2016152766
Cardiac Chemical Exchange Saturation Transfer MR Imaging Tracking of Cell Survival or Rejection in Mouse Models of Cell Therapy
Abstract
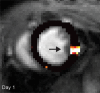
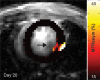
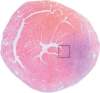
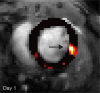
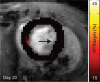
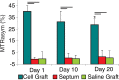
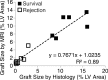
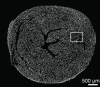
Purpose To examine whether cardiac chemical exchange saturation transfer (CEST) imaging can be serially and noninvasively used to probe cell survival or rejection after intramyocardial implantation in mice. Materials and Methods Experiments were compliant with the National Institutes of Health Guidelines on the Use of Laboratory Animals and approved by the Institutional Animal Care and Use Committee. One million C2C12 cells labeled with either europium (Eu) 10-(2-hydroxypropyl)-1,4,7-tetraazacyclododecane-1,4,7-triacetic acid (HP-DO3A) or saline via the hypotonic swelling technique were implanted into the anterior-lateral left ventricular wall in C57BL/6J (allogeneic model, n = 17) and C3H (syngeneic model, n = 13) mice. Imaging (frequency offsets of ±15 parts per million) was performed 1, 10, and 20 days after implantation, with the asymmetrical magnetization transfer ratio (MTRasym) calculated from image pairs. Histologic examination was performed at the conclusion of imaging. Changes in MTRasym over time and between mice were assessed by using two-way repeated-measures analysis of variance. Results MTRasym was significantly higher in C3H and C57BL/6J mice in grafts of Eu-HP-DO3A-labeled cells (40.2% ± 5.0 vs 37.8% ± 7.0, respectively) compared with surrounding tissue (-0.67% ± 1.7 vs -1.8% ± 5.3, respectively; P < .001) and saline-labeled grafts (-0.4% ± 6.0 vs -1.2% ± 3.6, respectively; P < .001) at day 1. In C3H mice, MTRasym remained increased (31.3% ± 9.2 on day 10, 28.7% ± 5.2 on day 20; P < .001 vs septum) in areas of in Eu-HP-DO3A-labeled cell grafts. In C57BL/6J mice, corresponding MTRasym values (11.3% ± 8.1 on day 10, 5.1% ± 9.4 on day 20; P < .001 vs day 1) were similar to surrounding myocardium by day 20 (P = .409). Histologic findings confirmed cell rejection in C57BL/6J mice. Estimation of graft area was similar with cardiac CEST imaging and histologic examination (R2 = 0.89). Conclusion Cardiac CEST imaging can be used to image cell survival and rejection in preclinical models of cell therapy. © RSNA, 2016 Online supplemental material is available for this article.
Figures




Similar articles
-
Advanced cardiac chemical exchange saturation transfer (cardioCEST) MRI for in vivo cell tracking and metabolic imaging.NMR Biomed. 2016 Jan;29(1):74-83. doi: 10.1002/nbm.3451. NMR Biomed. 2016. PMID: 26684053 Free PMC article.
-
Cardio-chemical exchange saturation transfer magnetic resonance imaging reveals molecular signatures of endogenous fibrosis and exogenous contrast media.Circ Cardiovasc Imaging. 2014 Dec 30;8(1):e002180. doi: 10.1161/CIRCIMAGING.114.002180. Print 2015 Jan. Circ Cardiovasc Imaging. 2014. PMID: 25550399
-
In Vivo 19F MR Imaging Cell Tracking of Inflammatory Macrophages and Site-specific Development of Colitis-associated Dysplasia.Radiology. 2017 Jan;282(1):194-201. doi: 10.1148/radiol.2016152387. Epub 2016 Jul 19. Radiology. 2017. PMID: 27440581 Free PMC article.
-
Creatine Chemical Exchange Saturation Transfer (Cr-CEST) Imaging Can Evaluate Cisplatin-induced Testicular Damage.Magn Reson Med Sci. 2023 Jul 1;22(3):345-351. doi: 10.2463/mrms.mp.2021-0125. Epub 2022 May 10. Magn Reson Med Sci. 2023. PMID: 35545506 Free PMC article.
-
Development of PET Imaging to Visualize Activated Macrophages Accumulated in the Transplanted iPSc-Derived Cardiac Myocytes of Allogeneic Origin for Detecting the Immune Rejection of Allogeneic Cell Transplants in Mice.PLoS One. 2016 Dec 8;11(12):e0165748. doi: 10.1371/journal.pone.0165748. eCollection 2016. PLoS One. 2016. PMID: 27930666 Free PMC article.
Cited by
-
Detection of tissue pH with quantitative chemical exchange saturation transfer magnetic resonance imaging.NMR Biomed. 2023 Jun;36(6):e4711. doi: 10.1002/nbm.4711. Epub 2022 Feb 20. NMR Biomed. 2023. PMID: 35141979 Free PMC article. Review.
-
The NIH Somatic Cell Genome Editing program.Nature. 2021 Apr;592(7853):195-204. doi: 10.1038/s41586-021-03191-1. Epub 2021 Apr 7. Nature. 2021. PMID: 33828315 Free PMC article. Review.
-
Chemistry of MRI Contrast Agents: Current Challenges and New Frontiers.Chem Rev. 2019 Jan 23;119(2):957-1057. doi: 10.1021/acs.chemrev.8b00363. Epub 2018 Oct 16. Chem Rev. 2019. PMID: 30350585 Free PMC article. Review.
-
A primer on in vivo cell tracking using MRI.Front Med (Lausanne). 2023 May 31;10:1193459. doi: 10.3389/fmed.2023.1193459. eCollection 2023. Front Med (Lausanne). 2023. PMID: 37324153 Free PMC article. Review.
-
Accelerated multi-target chemical exchange saturation transfer magnetic resonance imaging of the mouse heart.Phys Med Biol. 2021 Jul 16;66(14):10.1088/1361-6560/ac0e78. doi: 10.1088/1361-6560/ac0e78. Phys Med Biol. 2021. PMID: 34167100 Free PMC article.
References
-
- Wu JC. Molecular imaging: antidote to cardiac stem cell controversy. J Am Coll Cardiol 2008;52(20):1661–1664. - PubMed
-
- Pennell DJ. Cardiovascular magnetic resonance. Circulation 2010;121(5):692–705. - PubMed
-
- Frank JA, Miller BR, Arbab AS, et al. . Clinically applicable labeling of mammalian and stem cells by combining superparamagnetic iron oxides and transfection agents. Radiology 2003;228(2):480–487. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

