Safety and tolerability of the olaparib tablet formulation in Japanese patients with advanced solid tumours
- PMID: 27422301
- PMCID: PMC5010592
- DOI: 10.1007/s00280-016-3106-7
Safety and tolerability of the olaparib tablet formulation in Japanese patients with advanced solid tumours
Abstract
Purpose: This was the first Phase I study to assess the safety and tolerability of the tablet formulation of olaparib (Lynparza™), an oral poly(ADP-ribose) polymerase inhibitor, in Japanese patients with advanced solid tumours. The pharmacokinetic profile and antitumour activity of olaparib tablets were also assessed.
Methods: In this open-label, multicentre study (D081BC00001; NCT01813474), a single dose of olaparib (200 or 300 mg, tablets) was administered on day 1, followed 48 h afterwards by multiple dosing (200 or 300 mg twice daily [bid]) for 28-day cycles. Doses were escalated in successive cohorts, with an expansion cohort enrolled at the highest dose that was confirmed to be tolerable during dose escalation.
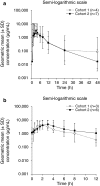
Results: Twenty-eight patients were enrolled and 23 were treated (n = 4, 7 and 12 at 200, 300 and 300 [expansion] mg bid, respectively). No patients experienced a dose-limiting toxicity, so the maximum tolerated dose was not defined. The most frequent adverse events were nausea (43.5 %), decreased appetite (30.4 %), anaemia (26.1 %) and constipation (26.1 %). No patient had dose reductions, two had dose interruptions, and two discontinued treatment because of adverse events. Absorption of olaparib was rapid following single and multiple dosing, and plasma concentrations declined biphasically after single dosing. No patients had a confirmed antitumour response.
Conclusions: Olaparib tablet doses of 200 and 300 mg bid were considered tolerable in Japanese patients with advanced solid tumours. Consistent with the global olaparib programme, 300 mg bid was selected as the recommended tablet dose for future studies.
Clinical trial registration number: NCT01813474.
Keywords: Clinical trial, Phase I; Olaparib; Poly(ADP-ribose) polymerase inhibitors; Safety; Solid tumours.
Conflict of interest statement
Kan Yonemori reports grants from AstraZeneca during the conduct of the study and personal fees from Eisai Co., Ltd, outside the submitted work. Kenji Tamura, Makoto Kodaira, Koshi Fujikawa, and Tamotsu Sagawa report personal fees from AstraZeneca during the conduct of the study. Taito Esaki reports grants from AstraZeneca during the conduct of the study, and outside of the submitted work: grants and personal fees from Boehringer Ingelheim, Eli Lilly, Merck Serono, and Taiho Pharmaceutical Co. Ltd; grants from Astellas, AstraZeneca, Bayer, Daiichi Sankyo, MSD, Novartis Pharma, Ono Pharmaceutical Co. Ltd, Sanofi, and Sumitomo Dainippon; and personal fees from Chugai and Yakult. Tsuyoshi Shirakawa reports personal fees from AstraZeneca during the conduct of the study. Fumihiko Hirai reports grants from AstraZeneca during the conduct of the study, and outside of the submitted work: grants and personal fees from Novartis Pharma; personal fees from Chugai Pharmaceutical Co., Ltd, Eli Lilly, Kyowa Hakko Kirin Co., Ltd, Ono Pharmaceutical Co., Ltd, and Taiho Pharmaceutical Co., Ltd. Yuki Yokoi, Toshio Kawata, Ben Hatano and Yasuo Takahashi report personal fees from AstraZeneca during the conduct of the study.
Figures
Similar articles
-
Pharmacokinetics and safety of olaparib tablets as monotherapy and in combination with paclitaxel: results of a Phase I study in Chinese patients with advanced solid tumours.Cancer Chemother Pharmacol. 2019 May;83(5):963-974. doi: 10.1007/s00280-019-03799-1. Epub 2019 Mar 18. Cancer Chemother Pharmacol. 2019. PMID: 30887180 Clinical Trial.
-
Trabectedin and olaparib in patients with advanced and non-resectable bone and soft-tissue sarcomas (TOMAS): an open-label, phase 1b study from the Italian Sarcoma Group.Lancet Oncol. 2018 Oct;19(10):1360-1371. doi: 10.1016/S1470-2045(18)30438-8. Epub 2018 Sep 11. Lancet Oncol. 2018. PMID: 30217671 Clinical Trial.
-
A Phase I, dose-finding and pharmacokinetic study of olaparib (AZD2281) in Japanese patients with advanced solid tumors.Cancer Sci. 2012 Mar;103(3):504-9. doi: 10.1111/j.1349-7006.2011.02179.x. Epub 2012 Jan 30. Cancer Sci. 2012. PMID: 22145984 Free PMC article. Clinical Trial.
-
Administration of the Tablet Formulation of Olaparib in Patients with Ovarian Cancer: Practical Guidance and Expectations.Oncologist. 2018 Jun;23(6):697-703. doi: 10.1634/theoncologist.2017-0485. Epub 2018 Mar 28. Oncologist. 2018. PMID: 29593098 Free PMC article. Review.
-
Olaparib Tablet: A Review in Ovarian Cancer Maintenance Therapy.Target Oncol. 2018 Dec;13(6):801-808. doi: 10.1007/s11523-018-0606-x. Target Oncol. 2018. PMID: 30456461 Review.
Cited by
-
Immunotherapy utilizing the combination of natural killer- and antibody dependent cellular cytotoxicity (ADCC)-mediating agents with poly (ADP-ribose) polymerase (PARP) inhibition.J Immunother Cancer. 2018 Nov 29;6(1):133. doi: 10.1186/s40425-018-0445-4. J Immunother Cancer. 2018. PMID: 30486888 Free PMC article.
-
Evaluation of Olaparib Tablet Safety and Pharmacokinetics in Healthy Chinese Male Subjects.Drug Des Devel Ther. 2024 Dec 3;18:5529-5539. doi: 10.2147/DDDT.S481481. eCollection 2024. Drug Des Devel Ther. 2024. PMID: 39650853 Free PMC article. Clinical Trial.
-
Bridging Olaparib Capsule and Tablet Formulations Using Population Pharmacokinetic Meta-analysis in Oncology Patients.Clin Pharmacokinet. 2019 May;58(5):615-625. doi: 10.1007/s40262-018-0714-x. Clin Pharmacokinet. 2019. PMID: 30357650
-
Phase I clinical study of oral olaparib in pediatric patients with refractory solid tumors: study protocol.BMC Pediatr. 2019 Jan 26;19(1):31. doi: 10.1186/s12887-019-1409-7. BMC Pediatr. 2019. PMID: 30684955 Free PMC article.
-
Counting the cost of public and philanthropic R&D funding: the case of olaparib.J Pharm Policy Pract. 2022 Aug 16;15(1):47. doi: 10.1186/s40545-022-00445-9. J Pharm Policy Pract. 2022. PMID: 35974344 Free PMC article.
References
-
- Evers B, Drost R, Schut E, de Bruin M, van der Burg E, Derksen PW, Holstege H, Liu X, van Drunen E, Beverloo HB, Smith GC, Martin NM, Lau A, O’Connor MJ, Jonkers J. Selective inhibition of BRCA2-deficient mammary tumor cell growth by AZD2281 and cisplatin. Clin Cancer Res. 2008;14:3916–3925. doi: 10.1158/1078-0432.CCR-07-4953. - DOI - PubMed
-
- Rottenberg S, Jaspers JE, Kersbergen A, van der Burg E, Nygren AO, Zander SA, Derksen PW, de Bruin M, Zevenhoven J, Lau A, Boulter R, Cranston A, O’Connor MJ, Martin NM, Borst P, Jonkers J. High sensitivity of BRCA1-deficient mammary tumors to the PARP inhibitor AZD2281 alone and in combination with platinum drugs. Proc Natl Acad Sci USA. 2008;105:17079–17084. doi: 10.1073/pnas.0806092105. - DOI - PMC - PubMed
-
- Fong PC, Boss DS, Yap TA, Tutt A, Wu P, Mergui-Roelvink M, Mortimer P, Swaisland H, Lau A, O’Connor MJ, Ashworth A, Carmichael J, Kaye SB, Schellens JHM, de Bono JS. Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. N Engl J Med. 2009;361:123–134. doi: 10.1056/NEJMoa0900212. - DOI - PubMed
-
- Audeh MW, Carmichael J, Penson RT, Friedlander M, Powell B, Bell-McGuinn KM, Scott C, Weitzel JN, Oaknin A, Loman N, Lu K, Schmutzler RK, Matulonis U, Wickens M, Tutt A. Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and recurrent ovarian cancer: a proof-of-concept trial. Lancet. 2010;376:245–251. doi: 10.1016/S0140-6736(10)60893-8. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources