A Revised Hemodynamic Theory of Age-Related Macular Degeneration
- PMID: 27423265
- PMCID: PMC4969140
- DOI: 10.1016/j.molmed.2016.06.009
A Revised Hemodynamic Theory of Age-Related Macular Degeneration
Abstract
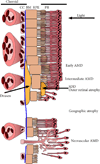
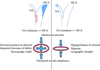
Age-related macular degeneration (AMD) afflicts one out of every 40 individuals worldwide, causing irreversible central blindness in millions. The transformation of various tissue layers within the macula in the retina has led to competing conceptual models of the molecular pathways, cell types, and tissues responsible for the onset and progression of AMD. A model that has persisted for over 6 decades is the hemodynamic, or vascular theory of AMD progression, which states that vascular dysfunction of the choroid underlies AMD pathogenesis. Here, we re-evaluate this hypothesis in light of recent advances on molecular, anatomic, and hemodynamic changes underlying choroidal dysfunction in AMD. We propose an updated, detailed model of hemodynamic dysfunction as a mechanism of AMD development and progression.
Copyright © 2016 Elsevier Ltd. All rights reserved.
Figures


References
-
- Wong WL, et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health. 2014;2(2):e106–e116. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

