Effects of D1 receptor knockout on fear and reward learning
- PMID: 27423521
- PMCID: PMC5001556
- DOI: 10.1016/j.nlm.2016.07.010
Effects of D1 receptor knockout on fear and reward learning
Abstract
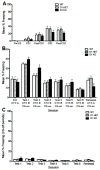
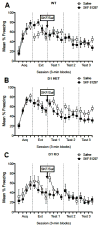
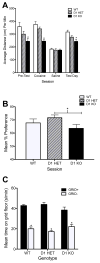
Dopamine signaling is involved in a variety of neurobiological processes that contribute to learning and memory. D1-like dopamine receptors (including D1 and D5 receptors) are thought to be involved in memory and reward processes, but pharmacological approaches have been limited in their ability to distinguish between D1 and D5 receptors. Here, we examine the effects of a specific knockout of D1 receptors in associative learning tasks involving aversive (shock) or appetitive (cocaine) unconditioned stimuli. We find that D1 knockout mice show similar levels of cued and contextual fear conditioning to WT controls following conditioning protocols involving one, two, or four shocks. D1 knockout mice show increased generalization of fear conditioning and extinction across contexts, revealed as increased freezing to a novel context following conditioning and decreased freezing to an extinguished cue during a contextual renewal test. Further, D1 knockout mice show mild enhancements in extinction following an injection of SKF81297, a D1/D5 receptor agonist, suggesting a role for D5 receptors in extinction enhancements induced by nonspecific pharmacological agonists. Finally, although D1 knockout mice show decreased locomotion induced by cocaine, they are able to form a cocaine-induced conditioned place preference. We discuss these findings in terms of the role of dopamine D1 receptors in general learning and memory processes.
Keywords: Cocaine; D1 receptor; D5 receptor; Dopamine; Extinction; Fear; Learning.
Copyright © 2016 Elsevier Inc. All rights reserved.
Figures
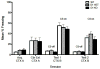
References
-
- Beaulieu JM, Gainetdinov RR. The physiology, signaling, and pharmacology of dopamine receptors. Pharmacol Rev. 2011;63(1):182–217. - PubMed
-
- Cervo L, Samanin R. Effects of dopaminergic and glutamatergic receptor antagonists on the acquisition and expression of cocaine conditioning place preference. Brain Res. 1995;673(2):242–50. - PubMed
-
- Cunningham CL, Gremel CM, Groblewski PA. Drug-induced conditioned place preference and aversion in mice. Nat Protoc. 2006;1(4):1662–70. - PubMed
-
- El-Ghundi M, O’Dowd BF, George SR. Prolonged fear responses in mice lacking dopamine D1 receptor. Brain Res. 2001;892(1):86–93. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

