Detection and quantification of epithelial progenitor cell populations in human healthy and IPF lungs
- PMID: 27423691
- PMCID: PMC4947297
- DOI: 10.1186/s12931-016-0404-x
Detection and quantification of epithelial progenitor cell populations in human healthy and IPF lungs
Abstract
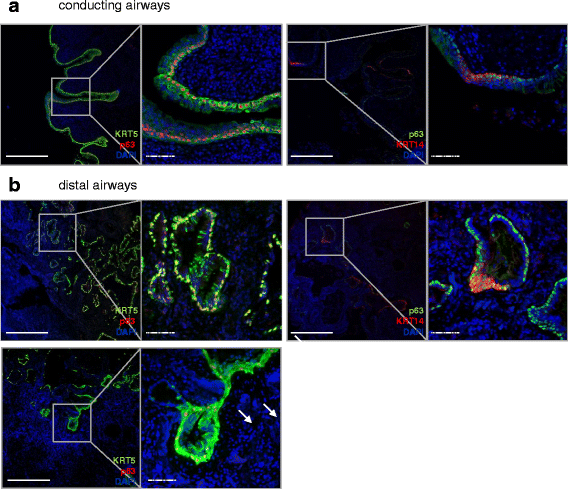
Background: In the human lung, epithelial progenitor cells in the airways give rise to the differentiated pseudostratified airway epithelium. In mice, emerging evidence confers a progenitor function to cytokeratin 5 (KRT5(+)) or cytokeratin 14 (KRT14(+))-positive basal cells of the airway epithelium. Little is known, however, about the distribution of progenitor subpopulations in the human lung, particularly about aberrant epithelial differentiation in lung disease, such as idiopathic pulmonary fibrosis (IPF).
Methods: Here, we used multi-color immunofluorescence analysis to detect and quantify the distribution of airway epithelial progenitor subpopulations in human lungs obtained from healthy donors or IPF patients.
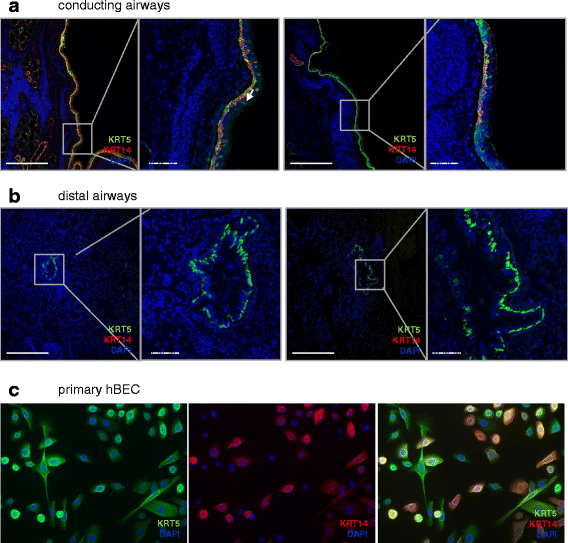
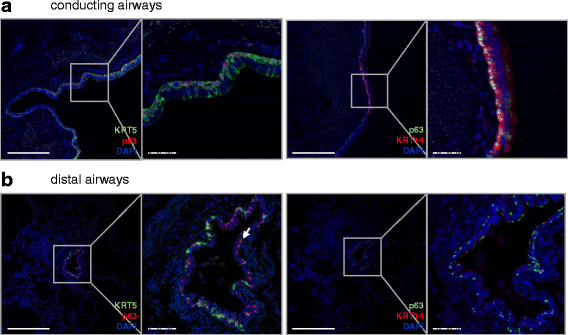
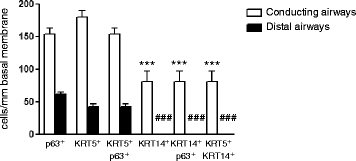
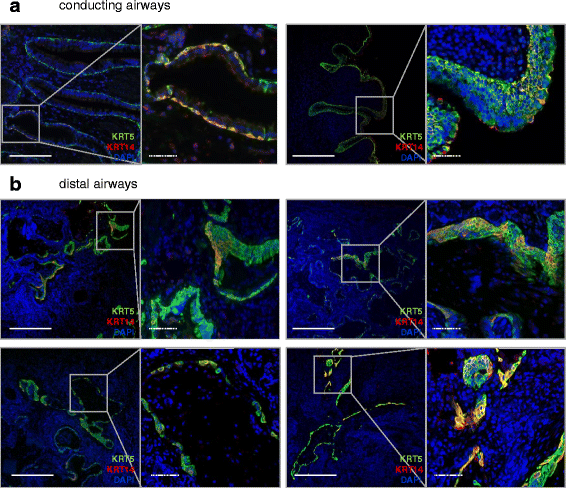
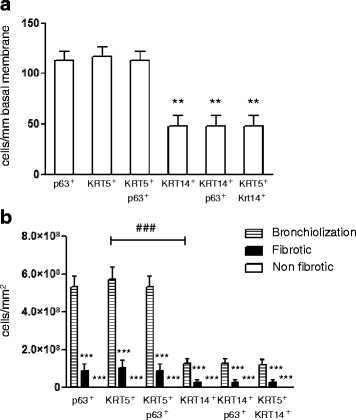
Results: In lungs from both, healthy donors and IPF patients, we detected KRT5(+)KRT14(-), KRT5(-)KRT14(+) and KRT5(+)KRT14(+) populations in the proximal airways. KRT14(+) cells, however, were absent in the distal airways of healthy lungs. In IPF, we detected a dramatic increase in the amount of KRT5(+) cells and the emergence of a frequent KRT5(+)KRT14(+) epithelial population, in particular in distal airways and alveolar regions. While the KRT14(-) progenitor population exhibited signs of proper epithelial differentiation, as evidenced by co-staining with pro-SPC, aquaporin 5, CC10, or MUC5B, the KRT14(+) cell population did not co-stain with bronchial/alveolar differentiation markers in IPF.
Conclusions: We provide, for the first time, a quantitative profile of the distribution of epithelial progenitor populations in human lungs. We show compelling evidence for dysregulation and aberrant differentiation of these populations in IPF.
Keywords: Basal cells; Epithelium; Human explants; Progenitor cells; Subpopulations.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

