Human adult neurogenesis across the ages: An immunohistochemical study
- PMID: 27424496
- PMCID: PMC5125837
- DOI: 10.1111/nan.12337
Human adult neurogenesis across the ages: An immunohistochemical study
Abstract
Aims: Neurogenesis in the postnatal human brain occurs in two neurogenic niches; the subventricular zone (SVZ) in the wall of the lateral ventricles and the subgranular zone (SGZ) of the hippocampus. The extent to which this physiological process continues into adulthood is an area of ongoing research. This study aimed to characterize markers of cell proliferation and assess the efficacy of antibodies used to identify neurogenesis in both neurogenic niches of the human brain.
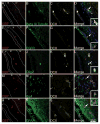
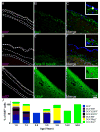
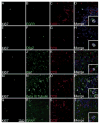
Methods: Cell proliferation and neurogenesis were simultaneously examined in the SVZ and SGZ of 23 individuals aged 0.2-59 years, using immunohistochemistry and immunofluorescence in combination with unbiased stereology.
Results: There was a marked decline in proliferating cells in both neurogenic niches in early infancy with levels reaching those seen in the adjacent parenchyma by 4 and 1 year of age, in the SVZ and SGZ, respectively. Furthermore, the phenotype of these proliferating cells in both niches changed with age. In infants, proliferating cells co-expressed neural progenitor (epidermal growth factor receptor), immature neuronal (doublecortin and beta III tubulin) and oligodendrocytic (Olig2) markers. However, after 3 years of age, microglia were the only proliferating cells found in either niche or in the adjacent parenchyma.
Conclusions: This study demonstrates a marked decline in neurogenesis in both neurogenic niches in early childhood, and that the sparse proliferating cells in the adult brain are largely microglia.
Keywords: adult neurogenesis; human post-mortem brain tissue; immunohistochemistry; microglial proliferation; subventricular zone and subgranular zone.
© 2016 British Neuropathological Society.
Conflict of interest statement
Statement of Conflict of Interest None of the authors have any conflict of interest in respect of the manuscript contents.
Figures




Comment in
-
Commentary on human adult neurogenesis across the ages: An immunohistochemical study.Neuropathol Appl Neurobiol. 2017 Aug;43(5):450-451. doi: 10.1111/nan.12377. Neuropathol Appl Neurobiol. 2017. PMID: 28039872 No abstract available.
-
Response to: Comment on 'Human adult neurogenesis across the ages: An immunohistochemical study'.Neuropathol Appl Neurobiol. 2017 Aug;43(5):452-454. doi: 10.1111/nan.12394. Neuropathol Appl Neurobiol. 2017. PMID: 28218954 No abstract available.
References
-
- Falk A, Frisen J. New neurons in old brains. Ann Med. 2005;37:480–6. - PubMed
-
- Mouret A, Murray K, Lledo PM. Centrifugal drive onto local inhibitory interneurons of the olfactory bulb. Ann N Y Acad Sci. 2009;1170:239–54. - PubMed
-
- Nissant A, Bardy C, Katagiri H, Murray K, Lledo PM. Adult neurogenesis promotes synaptic plasticity in the olfactory bulb. Nat Neurosci. 2009;12:728–30. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

