What Lies Beneath: Antibody Dependent Natural Killer Cell Activation by Antibodies to Internal Influenza Virus Proteins
- PMID: 27428437
- PMCID: PMC4919476
- DOI: 10.1016/j.ebiom.2016.04.029
What Lies Beneath: Antibody Dependent Natural Killer Cell Activation by Antibodies to Internal Influenza Virus Proteins
Abstract
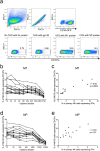
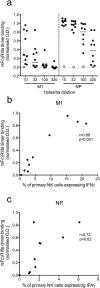
The conserved internal influenza proteins nucleoprotein (NP) and matrix 1 (M1) are well characterised for T cell immunity, but whether they also elicit functional antibodies capable of activating natural killer (NK) cells has not been explored. We studied NP and M1-specific ADCC activity using biochemical, NK cell activation and killing assays with plasma from healthy and influenza-infected subjects. Healthy adults had antibodies to M1 and NP capable of binding dimeric FcγRIIIa and activating NK cells. Natural symptomatic and experimental influenza infections resulted in a rise in antibody dependent NK cell activation post-infection to the hemagglutinin of the infecting strain, but changes in NK cell activation to M1 and NP were variable. Although antibody dependent killing of target cells infected with vaccinia viruses expressing internal influenza proteins was not detected, opsonising antibodies to NP and M1 likely contribute to an antiviral microenvironment by stimulating innate immune cells to secrete cytokines early in infection. We conclude that effector cell activating antibodies to conserved internal influenza proteins are common in healthy and influenza-infected adults. Given the significance of such antibodies in animal models of heterologous influenza infection, the definition of their importance and mechanism of action in human immunity to influenza is essential.
Keywords: Antibody dependent cellular cytotoxicity; Hemagglutinin; Influenza; Matrix protein 1; Natural killer cells; Nucleoprotein.
Copyright © 2016 The Authors. Published by Elsevier B.V. All rights reserved.
Figures









Comment in
-
"Universal Flu Vaccine": Can NK Cell-mediated ADCC Tip the Scales?EBioMedicine. 2016 Jun;8:18-19. doi: 10.1016/j.ebiom.2016.05.042. Epub 2016 Jun 3. EBioMedicine. 2016. PMID: 27428409 Free PMC article. No abstract available.
References
-
- Arnold J.N., Wormald M.R., Sim R.B., Rudd P.M., Dwek R.A. The impact of glycosylation on the biological function and structure of human immunoglobulins. Annu. Rev. Immunol. 2007;25:21–50. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

