Efficacy Projection of Obiltoxaximab for Treatment of Inhalational Anthrax across a Range of Disease Severity
- PMID: 27431222
- PMCID: PMC5038317
- DOI: 10.1128/AAC.00972-16
Efficacy Projection of Obiltoxaximab for Treatment of Inhalational Anthrax across a Range of Disease Severity
Abstract
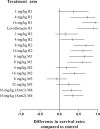
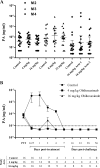
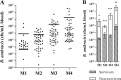
Inhalational anthrax has high mortality even with antibiotic treatment, and antitoxins are now recommended as an adjunct to standard antimicrobial regimens. The efficacy of obiltoxaximab, a monoclonal antibody against anthrax protective antigen (PA), was examined in multiple studies conducted in two animal models of inhalational anthrax. A single intravenous bolus of 1 to 32 mg/kg of body weight obiltoxaximab or placebo was administered to New Zealand White rabbits (two studies) and cynomolgus macaques (4 studies) at disease onset (significant body temperature increase or detection of serum PA) following lethal challenge with aerosolized Bacillus anthracis spores. The primary endpoint was survival. The relationship between efficacy and disease severity, defined by pretreatment bacteremia and toxemia levels, was explored. In rabbits, single doses of 1 to 16 mg/kg obiltoxaximab led to 17 to 93% survival. In two studies, survival following 16 mg/kg obiltoxaximab was 93% and 62% compared to 0% and 0% for placebo (P = 0.0010 and P = 0.0013, respectively). Across four macaque studies, survival was 6.3% to 78.6% following 4 to 32 mg/kg obiltoxaximab. In two macaque studies, 16 mg/kg obiltoxaximab reduced toxemia and led to survival rates of 31%, 35%, and 47% versus 0%, 0%, and 6.3% with placebo (P = 0.0085, P = 0.0053, P = 0.0068). Pretreatment bacteremia and toxemia levels inversely correlated with survival. Overall, obiltoxaximab monotherapy neutralized PA and increased survival across the range of disease severity, indicating clinical benefit of toxin neutralization with obiltoxaximab in both early and late stages of inhalational anthrax.
Copyright © 2016 Yamamoto et al.
Figures

Similar articles
-
Obiltoxaximab Prevents Disseminated Bacillus anthracis Infection and Improves Survival during Pre- and Postexposure Prophylaxis in Animal Models of Inhalational Anthrax.Antimicrob Agents Chemother. 2016 Sep 23;60(10):5796-805. doi: 10.1128/AAC.01102-16. Print 2016 Oct. Antimicrob Agents Chemother. 2016. PMID: 27431219 Free PMC article.
-
Development of Protective Immunity in New Zealand White Rabbits Challenged with Bacillus anthracis Spores and Treated with Antibiotics and Obiltoxaximab, a Monoclonal Antibody against Protective Antigen.Antimicrob Agents Chemother. 2018 Jan 25;62(2):e01590-17. doi: 10.1128/AAC.01590-17. Print 2018 Feb. Antimicrob Agents Chemother. 2018. PMID: 29133571 Free PMC article.
-
Obiltoxaximab: First Global Approval.Drugs. 2016 May;76(7):823-30. doi: 10.1007/s40265-016-0577-0. Drugs. 2016. PMID: 27085536 Review.
-
Obiltoxaximab: Adding to the Treatment Arsenal for Bacillus anthracis Infection.Ann Pharmacother. 2017 Oct;51(10):908-913. doi: 10.1177/1060028017713029. Epub 2017 Jun 2. Ann Pharmacother. 2017. PMID: 28573869 Review.
-
Animal-to-Human Dose Translation of Obiltoxaximab for Treatment of Inhalational Anthrax Under the US FDA Animal Rule.Clin Transl Sci. 2017 Jan;10(1):12-19. doi: 10.1111/cts.12433. Epub 2016 Dec 7. Clin Transl Sci. 2017. PMID: 27925405 Free PMC article.
Cited by
-
Pharmacokinetic and Pharmacodynamic Considerations for the Use of Monoclonal Antibodies in the Treatment of Bacterial Infections.Antibodies (Basel). 2018 Jan 4;7(1):5. doi: 10.3390/antib7010005. Antibodies (Basel). 2018. PMID: 31544858 Free PMC article. Review.
-
Safety, Pharmacokinetics, and Immunogenicity of Obiltoxaximab After Intramuscular Administration to Healthy Humans.Clin Pharmacol Drug Dev. 2018 Aug;7(6):652-660. doi: 10.1002/cpdd.410. Epub 2017 Nov 10. Clin Pharmacol Drug Dev. 2018. PMID: 29125719 Free PMC article. Clinical Trial.
-
Using Telemetry Data to Refine Endpoints for New Zealand White Rabbits Challenged with Bacillus anthracis.J Am Assoc Lab Anim Sci. 2017 Nov 1;56(6):792-801. J Am Assoc Lab Anim Sci. 2017. PMID: 29256375 Free PMC article.
-
Antitoxin Use in the Prevention and Treatment of Anthrax Disease: A Systematic Review.Clin Infect Dis. 2022 Oct 17;75(Suppl 3):S432-S440. doi: 10.1093/cid/ciac532. Clin Infect Dis. 2022. PMID: 36251559 Free PMC article.
-
Obiltoxaximab Prevents Disseminated Bacillus anthracis Infection and Improves Survival during Pre- and Postexposure Prophylaxis in Animal Models of Inhalational Anthrax.Antimicrob Agents Chemother. 2016 Sep 23;60(10):5796-805. doi: 10.1128/AAC.01102-16. Print 2016 Oct. Antimicrob Agents Chemother. 2016. PMID: 27431219 Free PMC article.
References
-
- Walsh JJ, Pesik N, Quinn CP, Urdaneta V, Dykewicz CA, Boyer AE, Guarner J, Wilkins P, Norville KJ, Barr JR, Zaki SR, Patel JB, Reagan SP, Pirkle JL, Treadwell TA, Messonnier NR, Rotz LD, Meyer RF, Stephens DS. 2007. A case of naturally acquired inhalation anthrax: clinical care and analyses of anti-protective antigen immunoglobulin G and lethal factor. Clin Infect Dis 44:968–971. doi:10.1086/512372. - DOI - PubMed
-
- Anaraki S, Addiman S, Nixon G, Krahe D, Ghosh R, Brooks T, Lloyd G, Spencer R, Walsh A, McCloskey B, Lightfoot N. 2008. Investigations and control measures following a case of inhalation anthrax in East London in a drum maker and drummer, October 2008. Euro Surveill 13:19076. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

