New anti-cancer chemicals Ertredin and its derivatives, regulate oxidative phosphorylation and glycolysis and suppress sphere formation in vitro and tumor growth in EGFRvIII-transformed cells
- PMID: 27431653
- PMCID: PMC4949881
- DOI: 10.1186/s12885-016-2521-9
New anti-cancer chemicals Ertredin and its derivatives, regulate oxidative phosphorylation and glycolysis and suppress sphere formation in vitro and tumor growth in EGFRvIII-transformed cells
Abstract
Background: EGFRvIII is a mutant form of the epidermal growth factor receptor gene (EGFR) that lacks exons 2-7. The resulting protein does not bind to ligands and is constitutively activated. The expression of EGFRvIII is likely confined to various types of cancer, particularly glioblastomas. Although an anti-EGFRvIII vaccine is of great interest, low-molecular-weight substances are needed to obtain better therapeutic efficacy. Thus, the purpose of this study is to identify low molecular weight substances that can suppress EGFRvIII-dependent transformation.
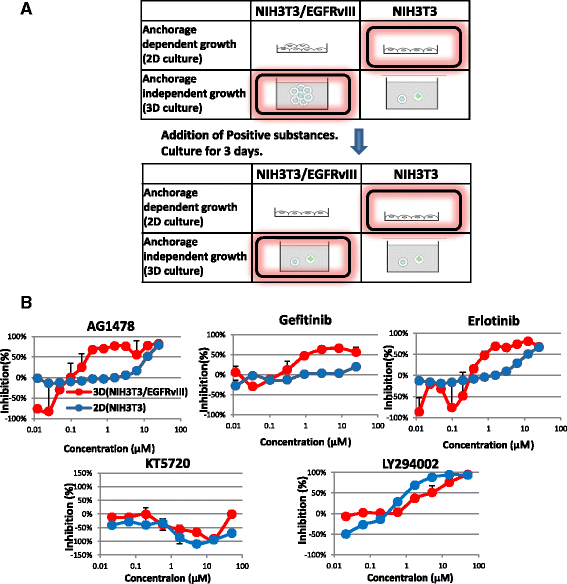
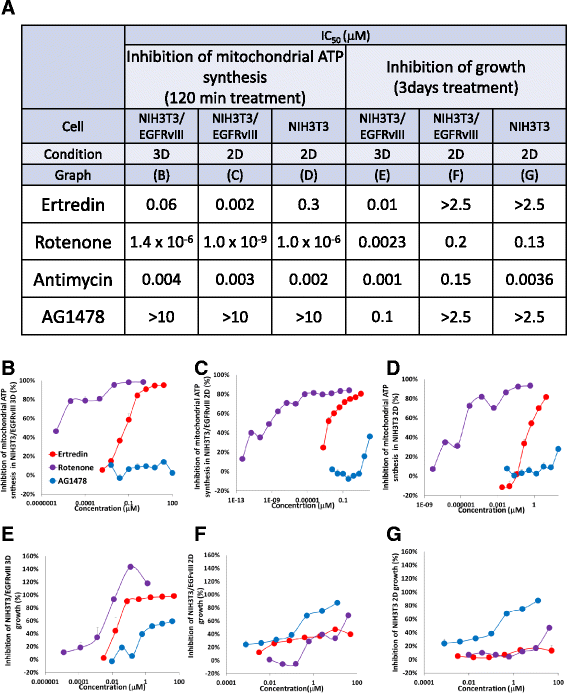
Methods: We constructed a new throughput screening system and searched for substances that decreased cell survival of NIH3T3/EGFRvIII spheres under 3-dimensional (3D)-culture conditions, but retained normal NIH3T3 cell growth under 2D-culture conditions. In vivo activity was examined using a mouse transplantation model, and derivatives were chemically synthesized. Functional characterization of the candidate molecules was investigated using an EGFR kinase assay, immunoprecipitation, western blotting, microarray analysis, quantitative polymerase chain reaction analysis, and measurement of lactate and ATP synthesis.
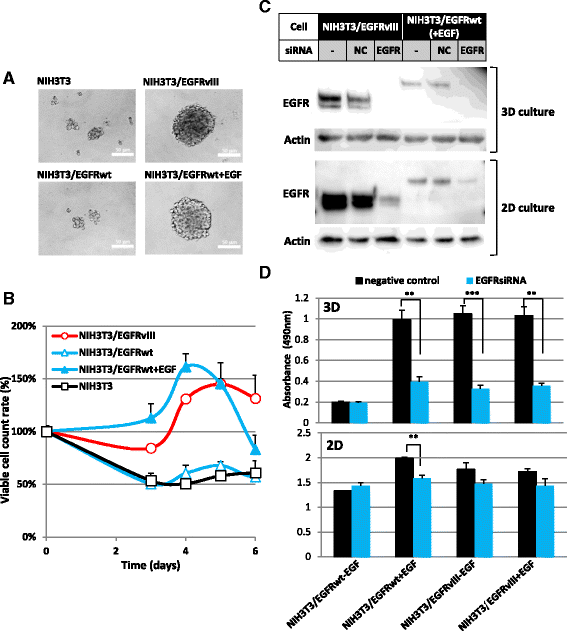
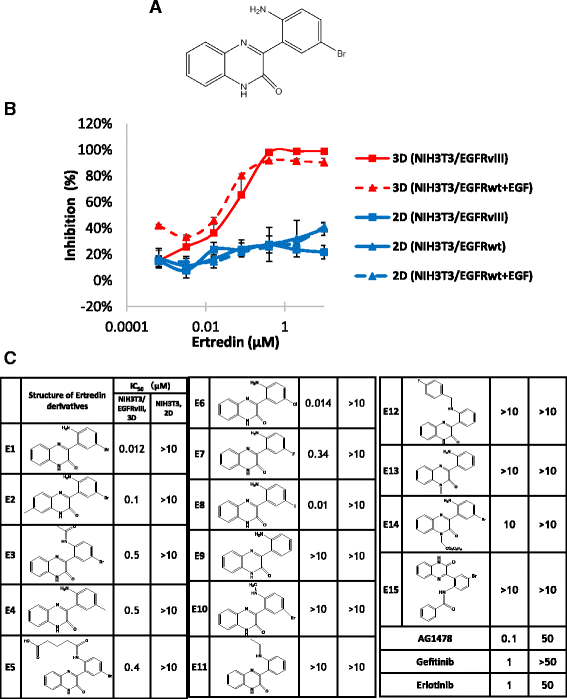
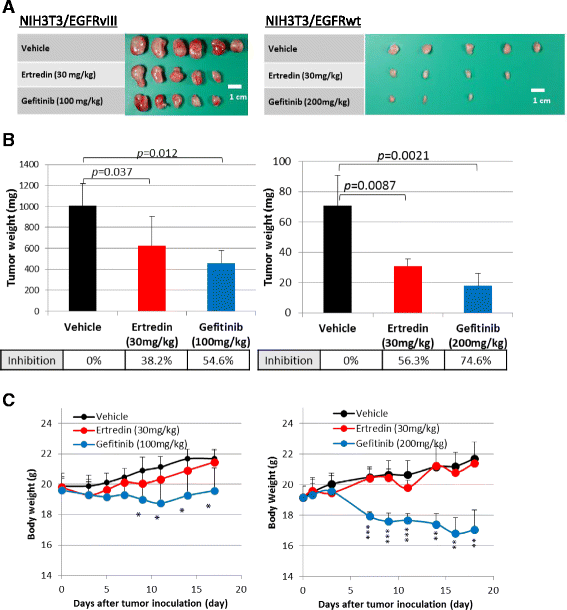
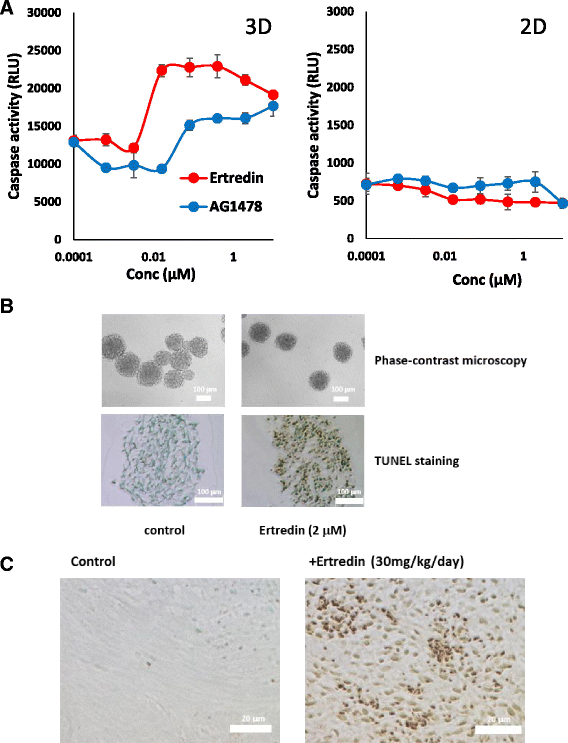
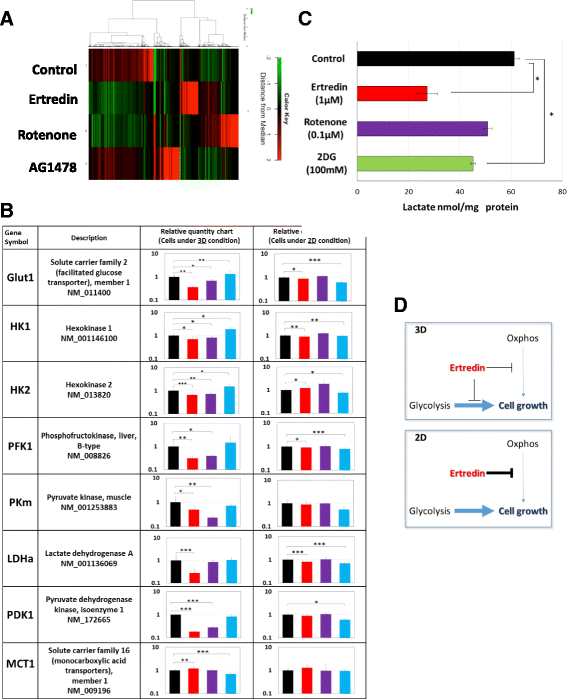
Results: In the course of screening 30,000 substances, a reagent, "Ertredin" was found to inhibit anchorage-independent 3D growth of sphere-forming cells transfected with EGFRvIII cDNA. Ertredin also inhibited sphere formation in cells expressing wild-type EGFR in the presence of EGF. However, it did not affect anchorage-dependent 2D growth of parental NIH3T3 cells. The 3D-growth-inhibitory activity of some derivatives, including those with new structures, was similar to Ertredin. Furthermore, we demonstrated that Ertredin suppressed tumor growth in an allograft transplantation mouse model injected with EGFRvIII- or wild-type EGFR-expressing cells; a clear toxicity to host animals was not observed. Functional characterization of Ertredin in cells expressing EGFRvIII indicated that it stimulated EGFRvIII ubiquitination, suppressed both oxidative phosphorylation and glycolysis under 3D conditions, and promoted cell apoptosis.
Conclusion: We developed a high throughput screening method based on anchorage-independent sphere formation induced by EGFRvIII-dependent transformation. In the course of screening, we identified Ertredin, which inhibited anchorage-independent 3D growth and tumor formation in nude mice. Functional analysis suggests that Ertredin suppresses both mitochondrial oxidative phosphorylation and cytosolic glycolysis in addition to promoting EGFRvIII degradation, and stimulates apoptosis in sphere-forming, EGFRvIII-overexpressing cells.
Keywords: 3D; Anchorage-independent; Apoptosis; EGFRvIII; Ertredin; Glycolysis; Oxidative phosphorylation; Sphere.
Figures

Similar articles
-
Enhanced Anticancer Activity of 7MeERT over Ertredin: A Comparative Study on Cancer Cell Proliferation and NDUFA12 Binding.Biomolecules. 2024 Sep 23;14(9):1197. doi: 10.3390/biom14091197. Biomolecules. 2024. PMID: 39334963 Free PMC article.
-
Epidermal growth factor receptor vIII enhances tumorigenicity in human breast cancer.Cancer Res. 2000 Jun 1;60(11):3081-7. Cancer Res. 2000. PMID: 10850460
-
NDUFA12 as a Functional Target of the Anticancer Compound Ertredin in Human Hepatoma Cells As Revealed by Label-Free Chemical Proteomics.J Proteome Res. 2024 Jan 5;23(1):130-141. doi: 10.1021/acs.jproteome.3c00471. Epub 2023 Nov 21. J Proteome Res. 2024. PMID: 38104258
-
Energy Metabolism Behavior and Response to Microenvironmental Factors of the Experimental Cancer Cell Models Differ from that of Actual Human Tumors.Mini Rev Med Chem. 2025;25(4):319-339. doi: 10.2174/0113895575322436240924101642. Mini Rev Med Chem. 2025. PMID: 39411957 Review.
-
Mutagenesis, tumorigenicity, and apoptosis: are the mitochondria involved?Mutat Res. 1998 Feb 26;398(1-2):19-26. doi: 10.1016/s0027-5107(97)00223-6. Mutat Res. 1998. PMID: 9626961 Review.
Cited by
-
Enhanced Anticancer Activity of 7MeERT over Ertredin: A Comparative Study on Cancer Cell Proliferation and NDUFA12 Binding.Biomolecules. 2024 Sep 23;14(9):1197. doi: 10.3390/biom14091197. Biomolecules. 2024. PMID: 39334963 Free PMC article.
-
Novel approaches for identification of anti-tumor drugs and new bioactive compounds.J Antibiot (Tokyo). 2017 Aug 30. doi: 10.1038/ja.2017.97. Online ahead of print. J Antibiot (Tokyo). 2017. PMID: 28852178 Review.
References
-
- Sebastian S, Settleman J, Reshkin SJ, Azzariti A, Bellizzi A, Paradiso A. The complexity of targeting EGFR signaling in cancer: from expression to turnover. Biochim Biophys Acta. 2006;1766(1):120–139. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

