Focal adhesions control cleavage furrow shape and spindle tilt during mitosis
- PMID: 27432211
- PMCID: PMC4949487
- DOI: 10.1038/srep29846
Focal adhesions control cleavage furrow shape and spindle tilt during mitosis
Abstract
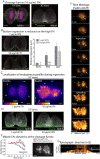
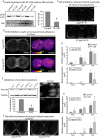
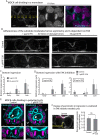
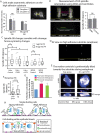
The geometry of the cleavage furrow during mitosis is often asymmetric in vivo and plays a critical role in stem cell differentiation and the relative positioning of daughter cells during development. Early observations of adhesive cell lines revealed asymmetry in the shape of the cleavage furrow, where the bottom (i.e., substrate attached side) of the cleavage furrow ingressed less than the top (i.e., unattached side). This data suggested substrate attachment could be regulating furrow ingression. Here we report a population of mitotic focal adhesions (FAs) controls the symmetry of the cleavage furrow. In single HeLa cells, stronger adhesion to the substrate directed less ingression from the bottom of the cell through a pathway including paxillin, focal adhesion kinase (FAK) and vinculin. Cell-cell contacts also direct ingression of the cleavage furrow in coordination with FAs in epithelial cells-MDCK-within monolayers and polarized cysts. In addition, mitotic FAs established 3D orientation of the mitotic spindle and the relative positioning of mother and daughter centrosomes. Therefore, our data reveals mitotic FAs as a key link between mitotic cell shape and spindle orientation, and may have important implications in our understanding stem cell homeostasis and tumorigenesis.
Figures
Similar articles
-
The novel actin/focal adhesion-associated protein MISP is involved in mitotic spindle positioning in human cells.Cell Cycle. 2013 May 1;12(9):1457-71. doi: 10.4161/cc.24602. Epub 2013 Apr 10. Cell Cycle. 2013. PMID: 23574715 Free PMC article.
-
Endomitotic megakaryocytes that form a bipolar spindle exhibit cleavage furrow ingression followed by furrow regression.Cell Cycle. 2007 Feb 15;6(4):455-60. doi: 10.4161/cc.6.4.3836. Epub 2007 Feb 8. Cell Cycle. 2007. PMID: 17312391
-
Specific detection of fission yeast primary septum reveals septum and cleavage furrow ingression during early anaphase independent of mitosis completion.PLoS Genet. 2018 May 29;14(5):e1007388. doi: 10.1371/journal.pgen.1007388. eCollection 2018 May. PLoS Genet. 2018. PMID: 29813053 Free PMC article.
-
Molecular pathways regulating mitotic spindle orientation in animal cells.Development. 2013 May;140(9):1843-56. doi: 10.1242/dev.087627. Development. 2013. PMID: 23571210 Free PMC article. Review.
-
Control of asymmetric cell division.Curr Opin Cell Biol. 2014 Dec;31:84-91. doi: 10.1016/j.ceb.2014.09.005. Epub 2014 Sep 28. Curr Opin Cell Biol. 2014. PMID: 25264944 Review.
Cited by
-
The balance between adhesion and contraction during cell division.Curr Opin Cell Biol. 2019 Feb;56:45-52. doi: 10.1016/j.ceb.2018.09.001. Epub 2018 Sep 28. Curr Opin Cell Biol. 2019. PMID: 30268802 Free PMC article. Review.
-
Spindle positioning and its impact on vertebrate tissue architecture and cell fate.Nat Rev Mol Cell Biol. 2021 Oct;22(10):691-708. doi: 10.1038/s41580-021-00384-4. Epub 2021 Jun 22. Nat Rev Mol Cell Biol. 2021. PMID: 34158639 Free PMC article. Review.
-
Coupling to substrate adhesions drives the maturation of muscle stress fibers into myofibrils within cardiomyocytes.Mol Biol Cell. 2020 Jun 1;31(12):1273-1288. doi: 10.1091/mbc.E19-11-0652. Epub 2020 Apr 8. Mol Biol Cell. 2020. PMID: 32267210 Free PMC article.
-
Connections between the cell cycle, cell adhesion and the cytoskeleton.Philos Trans R Soc Lond B Biol Sci. 2019 Aug 19;374(1779):20180227. doi: 10.1098/rstb.2018.0227. Epub 2019 Jul 1. Philos Trans R Soc Lond B Biol Sci. 2019. PMID: 31431178 Free PMC article. Review.
-
Interplay between the plasma membrane and cell-cell adhesion maintains epithelial identity for correct polarised cell divisions.J Cell Sci. 2024 Mar 1;137(5):jcs261701. doi: 10.1242/jcs.261701. Epub 2023 Nov 28. J Cell Sci. 2024. PMID: 37888135 Free PMC article.
References
-
- Gupton S. L. & Waterman-Storer C. M. Spatiotemporal feedback between actomyosin and focal-adhesion systems optimizes rapid cell migration. Cell 125, 1361–1374 (2006). - PubMed
-
- Mitchison T. J. Actin based motility on retraction fibers in mitotic PtK2 cells. Cell Motil Cytoskeleton 22, 135–151 (1992). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

