Spironolactone Prevents Endothelial Nitric Oxide Synthase Uncoupling and Vascular Dysfunction Induced by β-Adrenergic Overstimulation: Role of Perivascular Adipose Tissue
- PMID: 27432866
- PMCID: PMC4978608
- DOI: 10.1161/HYPERTENSIONAHA.116.07911
Spironolactone Prevents Endothelial Nitric Oxide Synthase Uncoupling and Vascular Dysfunction Induced by β-Adrenergic Overstimulation: Role of Perivascular Adipose Tissue
Abstract
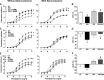
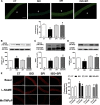
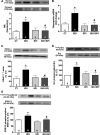
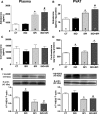
Sustained stimulation of β-adrenoceptors (β-ARs) and activation of renin-angiotensin-aldosterone system are common features of cardiovascular diseases with rising sympathetic activation, including essential hypertension, myocardial infarction, and heart failure. In this study, we investigated the role of AT1 receptor and mineralocorticoid receptor (MR) in the vascular alterations caused by β-AR overstimulation. β-AR overstimulation with associated cardiac hypertrophy and increased vasoconstrictor response to phenylephrine in aorta were modeled in rats by 7-day isoproterenol treatment. The increased vasoconstrictor response to phenylephrine in this model was blunted by the MR antagonist spironolactone, but not by the AT1 receptor antagonist losartan, despite the blunting of cardiac hypertrophy with both drugs. Spironolactone, but not losartan, restored NO bioavailability in association with lower endothelial nitric oxide synthase-derived superoxide production, increased endothelial nitric oxide synthase dimerization, and aortic HSP90 upregulation. MR genomic and nongenomic functions were activated in aortas from isoproterenol-treated rats. Isoproterenol did not modify plasma levels of MR ligands aldosterone and corticosterone but rather increased perivascular adipose tissue-derived corticosterone in association with increased expression of 11β-hydroxysteroid dehydrogenase type 1. The anticontractile effect of aortic perivascular adipose tissue was impaired by β-AR overstimulation and restored by MR blockade. These results suggest that activation of vascular MR signaling contributes to the vascular dysfunction induced by β-AR overstimulation associated with endothelial nitric oxide synthase uncoupling. These findings reveal an additional explanation for the protective effects of MR antagonists in cardiovascular disorders with sympathetic activation.
Keywords: adipose tissue; aorta; losartan; mineralocorticoid receptor.
© 2016 The Authors.
Figures


Similar articles
-
AT1 and aldosterone receptors blockade prevents the chronic effect of nandrolone on the exercise-induced cardioprotection in perfused rat heart subjected to ischemia and reperfusion.Cardiovasc Drugs Ther. 2014 Apr;28(2):125-35. doi: 10.1007/s10557-013-6503-8. Cardiovasc Drugs Ther. 2014. PMID: 24258356
-
Spironolactone prevents alterations associated with cardiac hypertrophy produced by isoproterenol in rats: involvement of serum- and glucocorticoid-regulated kinase type 1.Exp Physiol. 2012 Jun;97(6):710-8. doi: 10.1113/expphysiol.2011.063230. Epub 2012 Feb 10. Exp Physiol. 2012. PMID: 22327331
-
Cooperative Role of Mineralocorticoid Receptor and Caveolin-1 in Regulating the Vascular Response to Low Nitric Oxide-High Angiotensin II-Induced Cardiovascular Injury.J Pharmacol Exp Ther. 2015 Oct;355(1):32-47. doi: 10.1124/jpet.115.226043. Epub 2015 Jul 16. J Pharmacol Exp Ther. 2015. PMID: 26183312 Free PMC article.
-
The mineralocorticoid receptor in endothelial physiology and disease: novel concepts in the understanding of erectile dysfunction.Curr Pharm Des. 2008;14(35):3749-57. doi: 10.2174/138161208786898743. Curr Pharm Des. 2008. PMID: 19128227 Review.
-
The role of mineralocorticoid receptor signaling in the cross-talk between adipose tissue and the vascular wall.Cardiovasc Res. 2017 Jul 1;113(9):1055-1063. doi: 10.1093/cvr/cvx097. Cardiovasc Res. 2017. PMID: 28838041 Free PMC article. Review.
Cited by
-
The role of perivascular adipose tissue in obesity-induced vascular dysfunction.Br J Pharmacol. 2017 Oct;174(20):3425-3442. doi: 10.1111/bph.13650. Epub 2016 Nov 17. Br J Pharmacol. 2017. PMID: 27761903 Free PMC article. Review.
-
Potential pitfalls in analyzing structural uncoupling of eNOS: aging is not associated with increased enzyme monomerization.Am J Physiol Heart Circ Physiol. 2019 Jan 1;316(1):H80-H88. doi: 10.1152/ajpheart.00506.2018. Epub 2018 Oct 5. Am J Physiol Heart Circ Physiol. 2019. PMID: 30289292 Free PMC article.
-
Cardioprotective Action of Ginkgo biloba Extract against Sustained β-Adrenergic Stimulation Occurs via Activation of M2/NO Pathway.Front Pharmacol. 2017 May 11;8:220. doi: 10.3389/fphar.2017.00220. eCollection 2017. Front Pharmacol. 2017. PMID: 28553225 Free PMC article.
-
11β-Hydroxysteroid Dehydrogenases and Hypertension in the Metabolic Syndrome.Curr Hypertens Rep. 2017 Nov 14;19(12):100. doi: 10.1007/s11906-017-0797-z. Curr Hypertens Rep. 2017. PMID: 29138984 Free PMC article. Review.
-
Cellular mechanisms underlying obesity-induced arterial stiffness.Am J Physiol Regul Integr Comp Physiol. 2018 Mar 1;314(3):R387-R398. doi: 10.1152/ajpregu.00235.2016. Epub 2017 Nov 22. Am J Physiol Regul Integr Comp Physiol. 2018. PMID: 29167167 Free PMC article. Review.
References
-
- Grassi G. Sympathetic neural activity in hypertension and related diseases. Am J Hypertens. 2010;23:1052–1060. doi: 10.1038/ajh.2010.154. - PubMed
-
- Kim N, Chung J, Kim E, Han J. Changes in the Ca2+-activated K+ channels of the coronary artery during left ventricular hypertrophy. Circ Res. 2003;93:541–547. doi: 10.1161/01.RES.0000090087.66390.F2. - PubMed
-
- Kim HK, Park WS, Warda M, Park SY, Ko EA, Kim MH, Jeong SH, Heo HJ, Choi TH, Hwang YW, Lee SI, Ko KS, Rhee BD, Kim N, Han J. Beta adrenergic overstimulation impaired vascular contractility via actin-cytoskeleton disorganization in rabbit cerebral artery. PLoS One. 2012;7:e43884. doi: 10.1371/journal.pone.0043884. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

