E3 ubiquitin ligase Cbl-b negatively regulates C-type lectin receptor-mediated antifungal innate immunity
- PMID: 27432944
- PMCID: PMC4986534
- DOI: 10.1084/jem.20151932
E3 ubiquitin ligase Cbl-b negatively regulates C-type lectin receptor-mediated antifungal innate immunity
Abstract
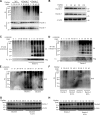
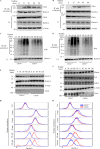
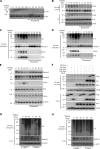
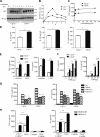
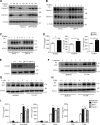
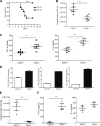
Activation of various C-type lectin receptors (CLRs) initiates potent proinflammatory responses against various microbial infections. However, how activated CLRs are negatively regulated remains unknown. In this study, we report that activation of CLRs Dectin-2 and Dectin-3 by fungi infections triggers them for ubiquitination and degradation in a Syk-dependent manner. Furthermore, we found that E3 ubiquitin ligase Casitas B-lineage lymphoma protein b (Cbl-b) mediates the ubiquitination of these activated CLRs through associating with each other via adapter protein FcR-γ and tyrosine kinase Syk, and then the ubiquitinated CLRs are sorted into lysosomes for degradation by an endosomal sorting complex required for transport (ESCRT) system. Therefore, the deficiency of either Cbl-b or ESCRT subunits significantly decreases the degradation of activated CLRs, thereby resulting in the higher expression of proinflammatory cytokines and inflammation. Consistently, Cbl-b-deficient mice are more resistant to fungi infections compared with wild-type controls. Together, our study indicates that Cbl-b negatively regulates CLR-mediated antifungal innate immunity, which provides molecular insight for designing antifungal therapeutic agents.
© 2016 Zhu et al.
Figures

Similar articles
-
Modulating Host Signaling Pathways to Promote Resistance to Infection by Candida albicans.Front Cell Infect Microbiol. 2017 Nov 21;7:481. doi: 10.3389/fcimb.2017.00481. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 29201860 Free PMC article. Review.
-
BLNK negatively regulates innate antifungal immunity through inhibiting c-Cbl-mediated macrophage migration.Proc Natl Acad Sci U S A. 2024 Oct 22;121(43):e2400920121. doi: 10.1073/pnas.2400920121. Epub 2024 Oct 16. Proc Natl Acad Sci U S A. 2024. PMID: 39413134 Free PMC article.
-
Targeting CBLB as a potential therapeutic approach for disseminated candidiasis.Nat Med. 2016 Aug;22(8):906-14. doi: 10.1038/nm.4141. Epub 2016 Jul 18. Nat Med. 2016. PMID: 27428899 Free PMC article.
-
Inhibition of CBLB protects from lethal Candida albicans sepsis.Nat Med. 2016 Aug;22(8):915-23. doi: 10.1038/nm.4134. Epub 2016 Jul 18. Nat Med. 2016. PMID: 27428901 Free PMC article.
-
Regulation of immune responses by E3 ubiquitin ligase Cbl-b.Cell Immunol. 2019 Jun;340:103878. doi: 10.1016/j.cellimm.2018.11.002. Epub 2018 Nov 7. Cell Immunol. 2019. PMID: 30442330 Free PMC article. Review.
Cited by
-
Involvement of Cbl-b-mediated macrophage inactivation in insulin resistance.World J Diabetes. 2017 Mar 15;8(3):97-103. doi: 10.4239/wjd.v8.i3.97. World J Diabetes. 2017. PMID: 28344752 Free PMC article. Review.
-
The first line of defense: effector pathways of anti-fungal innate immunity.Curr Opin Microbiol. 2020 Dec;58:160-165. doi: 10.1016/j.mib.2020.10.003. Epub 2020 Nov 17. Curr Opin Microbiol. 2020. PMID: 33217703 Free PMC article. Review.
-
Antifungal immunity: advances in PRR recognition, adaptive responses, and immune-based therapies.Sci China Life Sci. 2025 Aug;68(8):2206-2224. doi: 10.1007/s11427-024-2835-y. Epub 2025 Mar 5. Sci China Life Sci. 2025. PMID: 40055278 Review.
-
Modulating Host Signaling Pathways to Promote Resistance to Infection by Candida albicans.Front Cell Infect Microbiol. 2017 Nov 21;7:481. doi: 10.3389/fcimb.2017.00481. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 29201860 Free PMC article. Review.
-
Glutathione Reductase Promotes Fungal Clearance and Suppresses Inflammation during Systemic Candida albicans Infection in Mice.J Immunol. 2019 Oct 15;203(8):2239-2251. doi: 10.4049/jimmunol.1701686. Epub 2019 Sep 9. J Immunol. 2019. PMID: 31501257 Free PMC article.
References
-
- Bi L., Gojestani S., Wu W., Hsu Y.M.S., Zhu J., Ariizumi K., and Lin X.. 2010. CARD9 mediates dectin-2-induced IκBα kinase ubiquitination leading to activation of NF-κB in response to stimulation by the hyphal form of Candida albicans. J. Biol. Chem. 285:25969–25977. 10.1074/jbc.M110.131300 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

