Extracellular vesicles and viruses: Are they close relatives?
- PMID: 27432966
- PMCID: PMC4995926
- DOI: 10.1073/pnas.1605146113
Extracellular vesicles and viruses: Are they close relatives?
Abstract
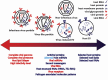
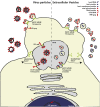
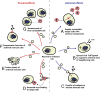
Extracellular vesicles (EVs) released by various cells are small phospholipid membrane-enclosed entities that can carry miRNA. They are now central to research in many fields of biology because they seem to constitute a new system of cell-cell communication. Physical and chemical characteristics of many EVs, as well as their biogenesis pathways, resemble those of retroviruses. Moreover, EVs generated by virus-infected cells can incorporate viral proteins and fragments of viral RNA, being thus indistinguishable from defective (noninfectious) retroviruses. EVs, depending on the proteins and genetic material incorporated in them, play a significant role in viral infection, both facilitating and suppressing it. Deciphering the mechanisms of EV-cell interactions may facilitate the design of EVs that inhibit viral infection and can be used as vehicles for targeted drug delivery.
Keywords: defective viruses; exosomes; extracellular vesicles; infection; viruses.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Colombo M, Raposo G, Théry C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol. 2014;30:255–289. - PubMed
-
- Valadi H, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9(6):654–659. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

