Assessing the performance of different DTI motion correction strategies in the presence of EPI distortion correction
- PMID: 27436169
- PMCID: PMC5118068
- DOI: 10.1002/hbm.23318
Assessing the performance of different DTI motion correction strategies in the presence of EPI distortion correction
Abstract
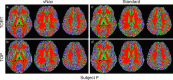
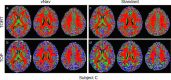
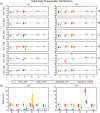
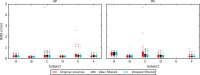
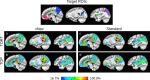
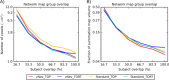
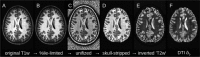
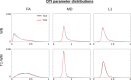
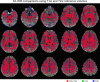
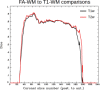
Diffusion tensor imaging (DTI) is susceptible to several artifacts due to eddy currents, echo planar imaging (EPI) distortion and subject motion. While several techniques correct for individual distortion effects, no optimal combination of DTI acquisition and processing has been determined. Here, the effects of several motion correction techniques are investigated while also correcting for EPI distortion: prospective correction, using navigation; retrospective correction, using two different popular packages (FSL and TORTOISE); and the combination of both methods. Data from a pediatric group that exhibited incidental motion in varying degrees are analyzed. Comparisons are carried while implementing eddy current and EPI distortion correction. DTI parameter distributions, white matter (WM) maps and probabilistic tractography are examined. The importance of prospective correction during data acquisition is demonstrated. In contrast to some previous studies, results also show that the inclusion of retrospective processing also improved ellipsoid fits and both the sensitivity and specificity of group tractographic results, even for navigated data. Matches with anatomical WM maps are highest throughout the brain for data that have been both navigated and processed using TORTOISE. The inclusion of both prospective and retrospective motion correction with EPI distortion correction is important for DTI analysis, particularly when studying subject populations that are prone to motion. Hum Brain Mapp 37:4405-4424, 2016. © 2016 Wiley Periodicals, Inc.
Keywords: diffusion tensor imaging; echo planar imaging volumetric navigator; fractional anisotropy; motion correction; tractography.
© 2016 Wiley Periodicals, Inc.
Figures




Similar articles
-
Motion artifact reduction in pediatric diffusion tensor imaging using fast prospective correction.J Magn Reson Imaging. 2015 May;41(5):1353-64. doi: 10.1002/jmri.24678. Epub 2014 Jun 17. J Magn Reson Imaging. 2015. PMID: 24935904 Free PMC article.
-
Real-time measurement and correction of both B0 changes and subject motion in diffusion tensor imaging using a double volumetric navigated (DvNav) sequence.Neuroimage. 2016 Feb 1;126:60-71. doi: 10.1016/j.neuroimage.2015.11.022. Epub 2015 Nov 14. Neuroimage. 2016. PMID: 26584865 Free PMC article.
-
Characterization and correction of diffusion gradient-induced eddy currents in second-order motion-compensated echo-planar and spiral cardiac DTI.Magn Reson Med. 2022 Dec;88(6):2378-2394. doi: 10.1002/mrm.29378. Epub 2022 Aug 2. Magn Reson Med. 2022. PMID: 35916545 Free PMC article.
-
Peripheral nerve diffusion tensor imaging: Overview, pitfalls, and future directions.J Magn Reson Imaging. 2018 May;47(5):1171-1189. doi: 10.1002/jmri.25876. Epub 2017 Oct 30. J Magn Reson Imaging. 2018. PMID: 29083521 Review.
-
Prospective motion correction in functional MRI.Neuroimage. 2017 Jul 1;154:33-42. doi: 10.1016/j.neuroimage.2016.11.014. Epub 2016 Nov 11. Neuroimage. 2017. PMID: 27845256 Free PMC article. Review.
Cited by
-
Towards an optimised processing pipeline for diffusion magnetic resonance imaging data: Effects of artefact corrections on diffusion metrics and their age associations in UK Biobank.Hum Brain Mapp. 2019 Oct 1;40(14):4146-4162. doi: 10.1002/hbm.24691. Epub 2019 Jun 7. Hum Brain Mapp. 2019. PMID: 31173439 Free PMC article.
-
What's new and what's next in diffusion MRI preprocessing.Neuroimage. 2022 Apr 1;249:118830. doi: 10.1016/j.neuroimage.2021.118830. Epub 2021 Dec 26. Neuroimage. 2022. PMID: 34965454 Free PMC article. Review.
-
Facilitating diffusion tensor imaging of the brain during continuous gross head motion with first and second order motion compensating diffusion gradients.Magn Reson Med. 2024 Apr;91(4):1556-1566. doi: 10.1002/mrm.29924. Epub 2023 Dec 10. Magn Reson Med. 2024. PMID: 38073070 Free PMC article.
-
Diffusion tensor imaging of the roots of the brachial plexus: a systematic review and meta-analysis of normative values.Clin Transl Imaging. 2020;8(6):419-431. doi: 10.1007/s40336-020-00393-x. Epub 2020 Oct 9. Clin Transl Imaging. 2020. PMID: 33282795 Free PMC article. Review.
-
White Matter Abnormalities in Children with HIV Infection and Exposure.Front Neuroanat. 2017 Sep 29;11:88. doi: 10.3389/fnana.2017.00088. eCollection 2017. Front Neuroanat. 2017. PMID: 29033797 Free PMC article.
References
-
- Andersson JL, Skare S, Ashburner J (2003): How to correct susceptibility distortions in spin‐echo echo‐planar images: Application to diffusion tensor imaging. Neuroimage 20:870–888. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

