Hypoxia-mediated impaired erythrocyte Lands' Cycle is pathogenic for sickle cell disease
- PMID: 27436223
- PMCID: PMC4951653
- DOI: 10.1038/srep29637
Hypoxia-mediated impaired erythrocyte Lands' Cycle is pathogenic for sickle cell disease
Abstract
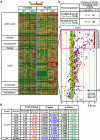
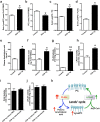
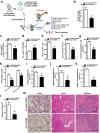
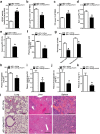
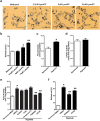
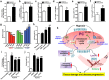
Although Lands' cycle was discovered in 1958, its function and cellular regulation in membrane homeostasis under physiological and pathological conditions remain largely unknown. Nonbiased high throughput metabolomic profiling revealed that Lands' cycle was impaired leading to significantly elevated erythrocyte membrane lysophosphatidylcholine (LysoPC) content and circulating and erythrocyte arachidonic acid (AA) in mice with sickle cell disease (SCD), a prevalent hemolytic genetic disorder. Correcting imbalanced Lands' cycle by knockdown of phospholipase 2 (cPLA2) or overexpression of lysophosphatidycholine acyltransferase 1 (LPCAT1), two key enzymes of Lands' cycle in hematopoietic stem cells, reduced elevated erythrocyte membrane LysoPC content and circulating AA levels and attenuated sickling, inflammation and tissue damage in SCD chimeras. Human translational studies validated SCD mouse findings and further demonstrated that imbalanced Lands' cycle induced LysoPC production directly promotes sickling in cultured mouse and human SCD erythrocytes. Mechanistically, we revealed that hypoxia-mediated ERK activation underlies imbalanced Lands' cycle by preferentially inducing the activity of PLA2 but not LPCAT in human and mouse SCD erythrocytes. Overall, our studies have identified a pathological role of imbalanced Lands' cycle in SCD erythrocytes, novel molecular basis regulating Lands' cycle and therapeutic opportunities for the disease.
Figures
Similar articles
-
Metabolomic and molecular insights into sickle cell disease and innovative therapies.Blood Adv. 2019 Apr 23;3(8):1347-1355. doi: 10.1182/bloodadvances.2018030619. Blood Adv. 2019. PMID: 31015210 Free PMC article. Review.
-
Lysophosphatidylcholine acyltransferase 3 knockdown-mediated liver lysophosphatidylcholine accumulation promotes very low density lipoprotein production by enhancing microsomal triglyceride transfer protein expression.J Biol Chem. 2012 Jun 8;287(24):20122-31. doi: 10.1074/jbc.M111.334664. Epub 2012 Apr 17. J Biol Chem. 2012. PMID: 22511767 Free PMC article.
-
Generation of membrane diversity by lysophospholipid acyltransferases.J Biochem. 2013 Jul;154(1):21-8. doi: 10.1093/jb/mvt048. Epub 2013 May 21. J Biochem. 2013. PMID: 23698096 Review.
-
Erythrocyte adaptive metabolic reprogramming under physiological and pathological hypoxia.Curr Opin Hematol. 2020 May;27(3):155-162. doi: 10.1097/MOH.0000000000000574. Curr Opin Hematol. 2020. PMID: 32141895 Free PMC article. Review.
-
Lysophosphatidylcholine acyltransferase 3 is the key enzyme for incorporating arachidonic acid into glycerophospholipids during adipocyte differentiation.Int J Mol Sci. 2012 Dec 3;13(12):16267-80. doi: 10.3390/ijms131216267. Int J Mol Sci. 2012. PMID: 23208369 Free PMC article.
Cited by
-
Fatty acid desaturase activity in mature red blood cells and implications for blood storage quality.Transfusion. 2021 Jun;61(6):1867-1883. doi: 10.1111/trf.16402. Epub 2021 Apr 26. Transfusion. 2021. PMID: 33904180 Free PMC article.
-
p53-driven lipidome influences non-cell-autonomous lysophospholipids in pancreatic cancer.Biol Direct. 2022 Mar 8;17(1):6. doi: 10.1186/s13062-022-00319-9. Biol Direct. 2022. PMID: 35255936 Free PMC article.
-
Red Blood Cell Metabolism in Pyruvate Kinase Deficient Patients.Front Physiol. 2021 Oct 21;12:735543. doi: 10.3389/fphys.2021.735543. eCollection 2021. Front Physiol. 2021. PMID: 34744776 Free PMC article.
-
Arachidonic Acid Metabolism Pathway Is Not Only Dominant in Metabolic Modulation but Associated With Phenotypic Variation After Acute Hypoxia Exposure.Front Physiol. 2018 Mar 16;9:236. doi: 10.3389/fphys.2018.00236. eCollection 2018. Front Physiol. 2018. PMID: 29615930 Free PMC article.
-
Neuronal transient receptor potential (TRP) channels and noxious sensory detection in sickle cell disease.Neurosci Lett. 2019 Feb 16;694:184-191. doi: 10.1016/j.neulet.2018.11.056. Epub 2018 Nov 30. Neurosci Lett. 2019. PMID: 30508569 Free PMC article. Review.
References
-
- Lands W. E. Metabolism of glycerolipides; a comparison of lecithin and triglyceride synthesis. J Biol Chem 231, 883–888 (1958). - PubMed
-
- Renooij W., Van Golde L. M., Zwaal R. F., Roelofsen B. & Van Deenen L. L. Preferential incorporation of fatty acids at the inside of human erythrocyte membranes. Biochim Biophys Acta 363, 287–292 (1974). - PubMed
-
- Connor W. E. et al.. Abnormal phospholipid molecular species of erythrocytes in sickle cell anemia. J Lipid Res 38, 2516–2528 (1997). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

