Spatially patterned matrix elasticity directs stem cell fate
- PMID: 27436901
- PMCID: PMC4978284
- DOI: 10.1073/pnas.1609731113
Spatially patterned matrix elasticity directs stem cell fate
Abstract
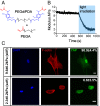
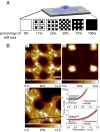
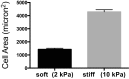
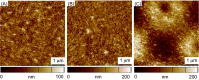
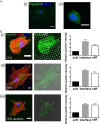
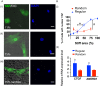
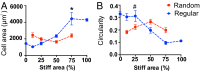
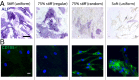
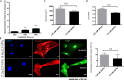
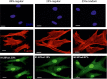
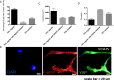
There is a growing appreciation for the functional role of matrix mechanics in regulating stem cell self-renewal and differentiation processes. However, it is largely unknown how subcellular, spatial mechanical variations in the local extracellular environment mediate intracellular signal transduction and direct cell fate. Here, the effect of spatial distribution, magnitude, and organization of subcellular matrix mechanical properties on human mesenchymal stem cell (hMSCs) function was investigated. Exploiting a photodegradation reaction, a hydrogel cell culture substrate was fabricated with regions of spatially varied and distinct mechanical properties, which were subsequently mapped and quantified by atomic force microscopy (AFM). The variations in the underlying matrix mechanics were found to regulate cellular adhesion and transcriptional events. Highly spread, elongated morphologies and higher Yes-associated protein (YAP) activation were observed in hMSCs seeded on hydrogels with higher concentrations of stiff regions in a dose-dependent manner. However, when the spatial organization of the mechanically stiff regions was altered from a regular to randomized pattern, lower levels of YAP activation with smaller and more rounded cell morphologies were induced in hMSCs. We infer from these results that irregular, disorganized variations in matrix mechanics, compared with regular patterns, appear to disrupt actin organization, and lead to different cell fates; this was verified by observations of lower alkaline phosphatase (ALP) activity and higher expression of CD105, a stem cell marker, in hMSCs in random versus regular patterns of mechanical properties. Collectively, this material platform has allowed innovative experiments to elucidate a novel spatial mechanical dosing mechanism that correlates to both the magnitude and organization of spatial stiffness.
Keywords: human mesenchymal stem cell; photodegradable hydrogel; spatial matrix stiffness.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Engler AJ, Sen S, Sweeney HL, Discher DE. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126(4):677–689. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

