Intracoronary Gene Transfer of Adenylyl Cyclase 6 in Patients With Heart Failure: A Randomized Clinical Trial
- PMID: 27437887
- PMCID: PMC5535743
- DOI: 10.1001/jamacardio.2016.0008
Intracoronary Gene Transfer of Adenylyl Cyclase 6 in Patients With Heart Failure: A Randomized Clinical Trial
Abstract
Importance: Gene transfer has rarely been tested in randomized clinical trials.
Objective: To evaluate the safety and efficacy of intracoronary delivery of adenovirus 5 encoding adenylyl cyclase 6 (Ad5.hAC6) in heart failure.
Design, setting, and participants: A randomized, double-blind, placebo-controlled, phase 2 clinical trial was conducted in US medical centers (randomization occurred from July 19, 2010, to October 30, 2014). Participants 18 to 80 years with symptomatic heart failure (ischemic and nonischemic) and an ejection fraction (EF) of 40% or less were screened; 86 individuals were enrolled, and 56 were randomized. Data analysis was of the intention-to-treat population. Participants underwent exercise testing and measurement of left ventricular EF (echocardiography) and then cardiac catheterization, where left ventricular pressure development (+dP/dt) and decline (-dP/dt) were recorded. Participants were randomized (3:1 ratio) to receive 1 of 5 doses of intracoronary Ad5.hAC6 or placebo. Participants underwent a second catheterization 4 weeks later for measurement of dP/dt. Exercise testing and EF were assessed 4 and 12 weeks after randomization.
Interventions: Intracoronary administration of Ad5.hAC6 (3.2 × 109 to 1012 virus particles) or placebo.
Main outcomes and measures: Primary end points included exercise duration and EF before and 4 and 12 weeks after randomization and peak rates of +dP/dt and -dP/dt before and 4 weeks after randomization. Fourteen placebo participants were compared (intention to treat) with 24 Ad5.hAC6 participants receiving the highest 2 doses (D4 + 5).
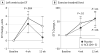
Results: Fifty-six individuals were randomized and monitored for up to 1 year. Forty-two participants (75%) received Ad5.hAC6 (mean [SE] age, 63 [1] years; EF, 30% [1%]), and 14 individuals (25%) received placebo (age, 62 [1] years; EF, 30% [2%]). Exercise duration showed no significant group differences (4 weeks, P = .27; 12 weeks, P = .47, respectively). The D4 + 5 participants had increased EF at 4 weeks (+6.0 [1.7] EF units; n = 21; P < .004), but not 12 weeks (+3.0 [2.4] EF units; n = 21; P = .16). Placebo participants showed no increase in EF at 4 weeks or 12 weeks. Exercise duration showed no between-group differences (4-week change from baseline: placebo, 27 [36] seconds; D4 + 5, 44 [25] seconds; P = .27; 12-week change from baseline: placebo, 44 [28] seconds; D4 + 5, 58 [29 seconds, P = .47). AC6 gene transfer increased basal left ventricular peak -dP/dt (4-week change from baseline: placebo, +93 [51] mm Hg/s; D4 + 5, -39 [33] mm Hg/s; placebo [n = 21]; P < .03); AC6 did not increase arrhythmias. The admission rate for patients with heart failure was 9.5% (4 of 42) in the AC6 group and 28.6% (4 of 14) in the placebo group (relative risk, 0.33 [95% CI, 0.08-1.36]; P = .10).
Conclusions and relevance: AC6 gene transfer safely increased LV function beyond standard heart failure therapy, attainable with one-time administration. Larger trials are warranted.
Trial registration: clinicaltrials.gov Identifier: NCT00787059.
Conflict of interest statement
Figures

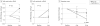
References
-
- Hall MJ, Levant S, DeFrances CJ. Hospitalization for Congestive Heart Failure: United States, 2000-2010. Hyattsville, MD: Centers for Disease Control and Prevention, National Center for Health Statistics; Oct, 2012. National Center for Health Statistics, Data Brief, No. 108.
-
- Sunahara RK, Dessauer CW, Gilman AG. Complexity and diversity of mammalian adenylyl cyclases. Annu Rev Pharmacol Toxicol. 1996;36:461–480. - PubMed
-
- Gao MH, Lai NC, Roth DM, et al. Adenylylcyclase increases responsiveness to catecholamine stimulation in transgenic mice. Circulation. 1999;99(12):1618–1622. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

