Prexasertib, a Chk1/Chk2 inhibitor, increases the effectiveness of conventional therapy in B-/T- cell progenitor acute lymphoblastic leukemia
- PMID: 27438145
- PMCID: PMC5288194
- DOI: 10.18632/oncotarget.10535
Prexasertib, a Chk1/Chk2 inhibitor, increases the effectiveness of conventional therapy in B-/T- cell progenitor acute lymphoblastic leukemia
Abstract
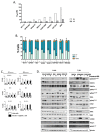
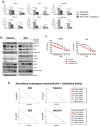
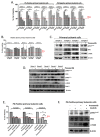
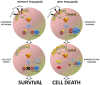
During the last few years many Checkpoint kinase 1/2 (Chk1/Chk2) inhibitors have been developed for the treatment of different type of cancers. In this study we evaluated the efficacy of the Chk 1/2 inhibitor prexasertib mesylate monohydrate in B-/T- cell progenitor acute lymphoblastic leukemia (ALL) as single agent and in combination with other drugs. The prexasertib reduced the cell viability in a dose and time dependent manner in all the treated cell lines. The cytotoxic activity was confirmed by the increment of apoptotic cells (Annexin V/Propidium Iodide staining), by the increase of γH2A.X protein expression and by the activation of different apoptotic markers (Parp-1 and pro-Caspase3 cleavage). Furthermore, the inhibition of Chk1 changed the cell cycle profile. In order to evaluate the chemo-sensitizer activity of the compound, different cell lines were treated for 24 and 48 hours with prexasertib in combination with other drugs (imatinib, dasatinib and clofarabine). The results from cell line models were strengthened in primary leukemic blasts isolated from peripheral blood of adult acute lymphoblastic leukemia patients. In this study we highlighted the mechanism of action and the effectiveness of prexasertib as single agent or in combination with other conventional drugs like imatinib, dasatinib and clofarabine in the treatment of B-/T-ALL.
Keywords: CHK1; DNA damage response; acute lymphoblastic leukemia; cell cycle; chemo-sensitizer agent.
Conflict of interest statement
GM has competing interests with Novartis, BMS, Roche, Pfizer, ARIAD, MSD.
Figures

Similar articles
-
Broad Spectrum Activity of the Checkpoint Kinase 1 Inhibitor Prexasertib as a Single Agent or Chemopotentiator Across a Range of Preclinical Pediatric Tumor Models.Clin Cancer Res. 2019 Apr 1;25(7):2278-2289. doi: 10.1158/1078-0432.CCR-18-2728. Epub 2018 Dec 18. Clin Cancer Res. 2019. PMID: 30563935 Free PMC article.
-
In vitro and in vivo single-agent efficacy of checkpoint kinase inhibition in acute lymphoblastic leukemia.J Hematol Oncol. 2015 Nov 5;8:125. doi: 10.1186/s13045-015-0206-5. J Hematol Oncol. 2015. PMID: 26542114 Free PMC article.
-
Prexasertib, a checkpoint kinase inhibitor: from preclinical data to clinical development.Cancer Chemother Pharmacol. 2020 Jan;85(1):9-20. doi: 10.1007/s00280-019-03950-y. Epub 2019 Sep 11. Cancer Chemother Pharmacol. 2020. PMID: 31512029 Review.
-
CHK1/2 Inhibitor Prexasertib Suppresses NOTCH Signaling and Enhances Cytotoxicity of Cisplatin and Radiation in Head and Neck Squamous Cell Carcinoma.Mol Cancer Ther. 2020 Jun;19(6):1279-1288. doi: 10.1158/1535-7163.MCT-19-0946. Epub 2020 May 5. Mol Cancer Ther. 2020. PMID: 32371584
-
Prexasertib: an investigational checkpoint kinase inhibitor for the treatment of high-grade serous ovarian cancer.Expert Opin Investig Drugs. 2020 Aug;29(8):779-792. doi: 10.1080/13543784.2020.1783238. Epub 2020 Jun 25. Expert Opin Investig Drugs. 2020. PMID: 32539469 Review.
Cited by
-
The DNA damage response pathway in normal hematopoiesis and malignancies.Int J Hematol. 2017 Sep;106(3):328-334. doi: 10.1007/s12185-017-2300-7. Epub 2017 Jul 13. Int J Hematol. 2017. PMID: 28707218 Review.
-
Preclinical efficacy of prexasertib in acute lymphoblastic leukemia.Br J Haematol. 2021 Sep;194(6):1094-1098. doi: 10.1111/bjh.17610. Epub 2021 Jun 7. Br J Haematol. 2021. PMID: 34096630 Free PMC article.
-
Targeting the PI3K/mTOR Pathway Augments CHK1 Inhibitor-Induced Replication Stress and Antitumor Activity in High-Grade Serous Ovarian Cancer.Cancer Res. 2020 Dec 1;80(23):5380-5392. doi: 10.1158/0008-5472.CAN-20-1439. Epub 2020 Sep 30. Cancer Res. 2020. PMID: 32998994 Free PMC article.
-
Broad Spectrum Activity of the Checkpoint Kinase 1 Inhibitor Prexasertib as a Single Agent or Chemopotentiator Across a Range of Preclinical Pediatric Tumor Models.Clin Cancer Res. 2019 Apr 1;25(7):2278-2289. doi: 10.1158/1078-0432.CCR-18-2728. Epub 2018 Dec 18. Clin Cancer Res. 2019. PMID: 30563935 Free PMC article.
-
Prexasertib treatment induces homologous recombination deficiency and synergizes with olaparib in triple-negative breast cancer cells.Breast Cancer Res. 2019 Sep 6;21(1):104. doi: 10.1186/s13058-019-1192-2. Breast Cancer Res. 2019. PMID: 31492187 Free PMC article.
References
-
- Moorman A V, Moorman A. New and emerging prognostic and predictive genetic biomarkers in B-cell precursor acute lymphoblastic leukemia. Hematology Am Soc Hematol Educ Program. 2015;9:7–16. - PubMed
-
- Foa R, Vitale A, Vignetti M, Meloni G, Guarini A, Propris MS De, et al. Dasatinib as first-line treatment for adult patients with Philadelphia chromosome – positive acute lymphoblastic leukemia. Therapy. 2011;118:6521–8. - PubMed
-
- Vignetti M, Fazi P, Cimino G, Martinelli G, Di Raimondo F, Ferrara F, et al. Imatinib plus steroids induces complete remissions and prolonged survival in elderly Philadelphia chromosome-positive patients with acute lymphoblastic leukemia without additional chemotherapy: Results of the Gruppo Italiano Malattie Ematologiche dell'Adu. Blood. 2007;109:3676–8. - PubMed
-
- Tamura K. Development of cell-cycle checkpoint therapy for solid tumors. Japanese Journal of Clinical Oncology. 2015:1097–102. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

