Human Cathelicidin Compensates for the Role of Apolipoproteins in Hepatitis C Virus Infectious Particle Formation
- PMID: 27440892
- PMCID: PMC5021414
- DOI: 10.1128/JVI.00471-16
Human Cathelicidin Compensates for the Role of Apolipoproteins in Hepatitis C Virus Infectious Particle Formation
Abstract
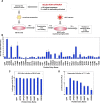
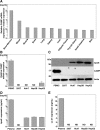
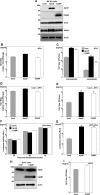
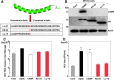
Exchangeable apolipoproteins (ApoA, -C, and -E) have been shown to redundantly participate in the formation of infectious hepatitis C virus (HCV) particles during the assembly process, although their precise role in the viral life cycle is not well understood. Recently, it was shown that the exogenous expression of only short sequences containing amphipathic α-helices from various apolipoproteins is sufficient to restore the formation of infectious HCV particles in ApoB and ApoE double-gene-knockout Huh7 (BE-KO) cells. In this study, through the expression of a small library of human secretory proteins containing amphipathic α-helix structures, we identified the human cathelicidin antimicrobial peptide (CAMP), the only known member of the cathelicidin family of antimicrobial peptides (AMPs) in humans and expressed mainly in bone marrow and leukocytes. We showed that CAMP is able to rescue HCV infectious particle formation in BE-KO cells. In addition, we revealed that the LL-37 domain in CAMP containing amphipathic α-helices is crucial for the compensation of infectivity in BE-KO cells, and the expression of CAMP in nonhepatic 293T cells expressing claudin 1 and microRNA miR-122 confers complete propagation of HCV. These results suggest the possibility of extrahepatic propagation of HCV in cells with low-level or no expression of apolipoproteins but expressing secretory proteins containing amphipathic α-helices such as CAMP.
Importance: Various exchangeable apolipoproteins play a pivotal role in the formation of infectious HCV during the assembly of viral particles, and amphipathic α-helix motifs in the apolipoproteins have been shown to be a key factor. To the best of our knowledge, we have identified for the first time the human cathelicidin CAMP as a cellular protein that can compensate for the role of apolipoproteins in the life cycle of HCV. We have also identified the domain in CAMP that contains amphipathic α-helices crucial for compensation and show that the expression of CAMP in nonhepatic cells expressing claudin 1 and miR-122 confers complete propagation of HCV. We speculate that low levels of HCV propagation might be possible in extrahepatic tissues expressing secretory proteins containing amphipathic α-helices without the expression of apolipoproteins.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures




References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

