Cell Wall Ultrastructure of Stem Wood, Roots, and Needles of a Conifer Varies in Response to Moisture Availability
- PMID: 27446114
- PMCID: PMC4919352
- DOI: 10.3389/fpls.2016.00882
Cell Wall Ultrastructure of Stem Wood, Roots, and Needles of a Conifer Varies in Response to Moisture Availability
Abstract
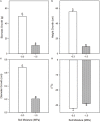
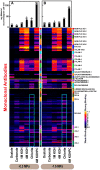
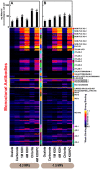
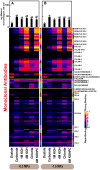
The composition, integrity, and architecture of the macromolecular matrix of cell walls, collectively referred to as cell wall ultrastructure, exhibits variation across species and organs and among cell types within organs. Indirect approaches have suggested that modifications to cell wall ultrastructure occur in response to abiotic stress; however, modifications have not been directly observed. Glycome profiling was used to study cell wall ultrastructure by examining variation in composition and extractability of non-cellulosic glycans in cell walls of stem wood, roots, and needles of loblolly pine saplings exposed to high and low soil moisture. Soil moisture influenced physiological processes and the overall composition and extractability of cell wall components differed as a function of soil moisture treatments. The strongest response of cell wall ultrastructure to soil moisture was increased extractability of pectic backbone epitopes in the low soil moisture treatment. The higher abundance of these pectic backbone epitopes in the oxalate extract indicate that the loosening of cell wall pectic components could be associated with the release of pectic signals as a stress response. The increased extractability of pectic backbone epitopes in response to low soil moisture availability was more pronounced in stem wood than in roots or needles. Additional responses to low soil moisture availability were observed in lignin-associated carbohydrates released in chlorite extracts of stem wood, including an increased abundance of pectic arabinogalactan epitopes. Overall, these results indicate that cell walls of loblolly pine organs undergo changes in their ultrastructural composition and extractability as a response to soil moisture availability and that cell walls of the stem wood are more responsive to low soil moisture availability compared to cell walls of roots and needles. To our knowledge, this is the first direct evidence, delineated by glycomic analyses, that abiotic stress affects cell wall ultrastructure. This study is also unique in that glycome profiling of pine needles has never before been reported.
Keywords: Pinus taeda; cell walls; glycome profiling; moisture stress; monoclonal antibodies; pectin; xylan.
Figures
Similar articles
-
A quantitative method for analyzing glycome profiles of plant cell walls.Carbohydr Res. 2017 Aug 7;448:128-135. doi: 10.1016/j.carres.2017.06.009. Epub 2017 Jun 19. Carbohydr Res. 2017. PMID: 28662408
-
Differential expression of genes encoding cell wall proteins in vascular tissues from vertical and bent loblolly pine trees.Tree Physiol. 2000 Apr;20(7):457-466. doi: 10.1093/treephys/20.7.457. Tree Physiol. 2000. PMID: 12651441
-
Insights into plant cell wall structure, architecture, and integrity using glycome profiling of native and AFEXTM-pre-treated biomass.J Exp Bot. 2015 Jul;66(14):4279-94. doi: 10.1093/jxb/erv107. Epub 2015 Apr 23. J Exp Bot. 2015. PMID: 25911738 Free PMC article.
-
Immunological Approaches to Biomass Characterization and Utilization.Front Bioeng Biotechnol. 2015 Oct 28;3:173. doi: 10.3389/fbioe.2015.00173. eCollection 2015. Front Bioeng Biotechnol. 2015. PMID: 26579515 Free PMC article. Review.
-
Research Progress of Wood Cell Wall Modification and Functional Improvement: A Review.Materials (Basel). 2022 Feb 21;15(4):1598. doi: 10.3390/ma15041598. Materials (Basel). 2022. PMID: 35208141 Free PMC article. Review.
Cited by
-
ARABIDOPSIS DEHISCENCE ZONE POLYGALACTURONASE 1 (ADPG1) releases latent defense signals in stems with reduced lignin content.Proc Natl Acad Sci U S A. 2020 Feb 11;117(6):3281-3290. doi: 10.1073/pnas.1914422117. Epub 2020 Jan 23. Proc Natl Acad Sci U S A. 2020. PMID: 31974310 Free PMC article.
-
Importance of incorporating spatial and temporal variability of biomass yield and quality in bioenergy supply chain.Sci Rep. 2023 Apr 26;13(1):6813. doi: 10.1038/s41598-023-28671-4. Sci Rep. 2023. PMID: 37100831 Free PMC article.
-
Mechanistic insights into the digestion of complex dietary fibre by the rumen microbiota using combinatorial high-resolution glycomics and transcriptomic analyses.Comput Struct Biotechnol J. 2021 Dec 9;20:148-164. doi: 10.1016/j.csbj.2021.12.009. eCollection 2022. Comput Struct Biotechnol J. 2021. PMID: 34976318 Free PMC article.
-
Comparative transcriptomics indicate changes in cell wall organization and stress response in seedlings during spaceflight.Am J Bot. 2017 Aug;104(8):1219-1231. doi: 10.3732/ajb.1700079. Am J Bot. 2017. PMID: 28827451 Free PMC article.
References
-
- Aubrey D. P., Coyle D. R., Coleman M. D. (2012). Functional groups show distinct differences in nitrogen cycling during early stand development: implications for forest management. Plant Soil 351, 219–236. 10.1007/s11104-011-0946-0 - DOI
-
- Chartzoulakis K., Patakas A., Kofidis G., Bosabalidis A., Nastou A. (2002). Water stress affects leaf anatomy, gas exchange, water relations and growth of two avocado cultivars. Sci. Hortic. 95, 39–50. 10.1016/S0304-4238(02)00016-X - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources

