Dynamic full field optical coherence tomography: subcellular metabolic contrast revealed in tissues by interferometric signals temporal analysis
- PMID: 27446672
- PMCID: PMC4929658
- DOI: 10.1364/BOE.7.001511
Dynamic full field optical coherence tomography: subcellular metabolic contrast revealed in tissues by interferometric signals temporal analysis
Abstract
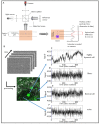
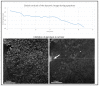
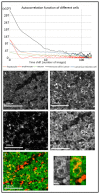
We developed a new endogenous approach to reveal subcellular metabolic contrast in fresh ex vivo tissues taking advantage of the time dependence of the full field optical coherence tomography interferometric signals. This method reveals signals linked with local activity of the endogenous scattering elements which can reveal cells where other OCT-based techniques fail or need exogenous contrast agents. We benefit from the micrometric transverse resolution of full field OCT to image intracellular features. We used this time dependence to identify different dynamics at the millisecond scale on a wide range of organs in normal or pathological conditions.
Keywords: (100.0100) Image processing; (110.4500) Optical coherence tomography; (110.6915) Time imaging; (170.1530) Cell analysis; (170.6930) Tissue; (180.6900) Three-dimensional microscopy.
Figures


References
-
- Alberts B., Johnson A., Lewis J., Morgan D., Raff M., Roberts K., Walter P., Molecular Biology of the Cell, 6th ed. (Garland Science, 2014).
-
- Guo M., Ehrlicher A. J., Jensen M. H., Renz M., Moore J. R., Goldman R. D., Lippincott-Schwartz J., Mackintosh F. C., Weitz D. A., “Probing the stochastic, motor-driven properties of the cytoplasm using force spectrum microscopy,” Cell 158(4), 822–832 (2014).10.1016/j.cell.2014.06.051 - DOI - PMC - PubMed
-
- Shaked N. T., Rinehart M. T., Wax A., “Quantitative phase microscopy of biological cell dynamics by wide-field digital interferometry,” in Coherent Light Microscopy (Springer, 2011), pp. 169–198.
LinkOut - more resources
Full Text Sources
Other Literature Sources
