Toxin-Antitoxin Systems in Clinical Pathogens
- PMID: 27447671
- PMCID: PMC4963858
- DOI: 10.3390/toxins8070227
Toxin-Antitoxin Systems in Clinical Pathogens
Abstract
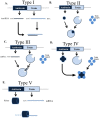
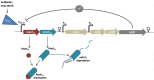
Toxin-antitoxin (TA) systems are prevalent in bacteria and archaea. Although not essential for normal cell growth, TA systems are implicated in multiple cellular functions associated with survival under stress conditions. Clinical strains of bacteria are currently causing major human health problems as a result of their multidrug resistance, persistence and strong pathogenicity. Here, we present a review of the TA systems described to date and their biological role in human pathogens belonging to the ESKAPE group (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa and Enterobacter spp.) and others of clinical relevance (Escherichia coli, Burkholderia spp., Streptococcus spp. and Mycobacterium tuberculosis). Better understanding of the mechanisms of action of TA systems will enable the development of new lines of treatment for infections caused by the above-mentioned pathogens.
Keywords: Toxin-Antitoxin; chromosome; clinical; pathogens; persistance; plasmids; resistance; virulence.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

