Integrin and dystroglycan compensate each other to mediate laminin-dependent basement membrane assembly and epiblast polarization
- PMID: 27449702
- PMCID: PMC5250580
- DOI: 10.1016/j.matbio.2016.07.005
Integrin and dystroglycan compensate each other to mediate laminin-dependent basement membrane assembly and epiblast polarization
Abstract
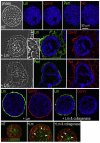
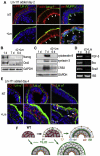
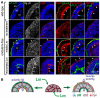
During early embryogenesis, endodermal γ1-laminin expression is required for basement membrane (BM) assembly, promoting conversion of non-polar pluripotent cells into polarized epiblast. The influence of laminin-111 (Lm111) and its integrin and dystroglycan (DG) receptors on epiblast in embryoid bodies (EBs), a model for differentiation of the embryonic plate, was further investigated. Lm111 added to the medium of EBs initiated conversion of inner nonpolar cell to the polarized epiblast epithelium with an exterior-to-central basal-to-apical orientation. Microinjection of Lm111 into EB interiors resulted in an interior BM with complete inversion of cell polarity. Lm111 assembled a BM on integrin-β1 null EBs with induction of polarization at reduced efficiency. β-Integrin compensation was not detected in these nulls with integrin adaptor proteins failing to assemble. A dimer of laminin LG domains 4-5 (LZE3) engineered to strongly bind to α-dystroglycan almost completely inhibited laminin accumulation on integrin β1-null EBs, reducing BM and ablating cell polarization. When Lm111 was incubated with integrin-β1/dystroglycan double-knockout EBs, laminin failed to accumulate on the EBs, the EBs did not differentiate, and the EBs underwent apoptosis. Collectively the findings support the hypotheses that the locus of laminin cell surface assembly can determine the axis of epithelial polarity. This requires integrin- and/or dystroglycan-dependent binding to laminin LG domains with the highest efficiency achieved when both receptors are present. Finally, EBs that cannot assemble a matrix undergo apoptosis.
Keywords: cell polarity; embryonic stem cell; endoderm; epiblast.
Copyright © 2017 The Authors. Published by Elsevier B.V. All rights reserved.
Figures



References
-
- Li S, Edgar D, Fassler R, Wadsworth W, Yurchenco PD. The role of laminin in embryonic cell polarization and tissue organization. Dev. Cell. 2003;4(5):613–624. - PubMed
-
- Wilkinson DG, Bhatt S, Herrmann BG. Expression pattern of the mouse T gene and its role in mesoderm formation. Nature. 1990;343(6259):657–659. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

