Distinct Metabolic Requirements of Exhausted and Functional Virus-Specific CD8 T Cells in the Same Host
- PMID: 27452473
- PMCID: PMC4977274
- DOI: 10.1016/j.celrep.2016.06.078
Distinct Metabolic Requirements of Exhausted and Functional Virus-Specific CD8 T Cells in the Same Host
Abstract
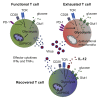
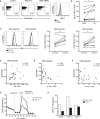
T cells undergo profound metabolic changes to meet the increased energy demands of maintaining an antiviral response. We postulated that differences in metabolic reprogramming would shape the efficacy of CD8 T cells mounted against persistent viral infections. We found that the poorly functional PD-1(hi) T cell response against hepatitis B virus (HBV) had upregulated the glucose transporter, Glut1, an effect recapitulated by oxygen deprivation to mimic the intrahepatic environment. Glut1(hi) HBV-specific T cells were dependent on glucose supplies, unlike the more functional cytomegalovirus (CMV)-specific T cells that could utilize oxidative phosphorylation in the absence of glucose. The inability of HBV-specific T cells to switch to oxidative phosphorylation was accompanied by increased mitochondrial size and lower mitochondrial potential, indicative of mitochondrial dysfunction. Interleukin (IL)-12, which recovers HBV-specific T cell effector function, increased their mitochondrial potential and reduced their dependence on glycolysis. Our findings suggest that mitochondrial defects limit the metabolic plasticity of exhausted HBV-specific T cells.
Copyright © 2016 The Author(s). Published by Elsevier Inc. All rights reserved.
Figures



References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

