Local Innate Responses to TLR Ligands in the Chicken Trachea
- PMID: 27455308
- PMCID: PMC4974541
- DOI: 10.3390/v8070207
Local Innate Responses to TLR Ligands in the Chicken Trachea
Abstract
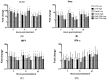
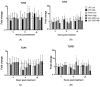
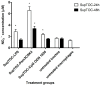
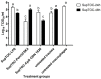
The chicken upper respiratory tract is the portal of entry for respiratory pathogens, such as avian influenza virus (AIV). The presence of microorganisms is sensed by pathogen recognition receptors (such as Toll-like receptors (TLRs)) of the innate immune defenses. Innate responses are essential for subsequent induction of potent adaptive immune responses, but little information is available about innate antiviral responses of the chicken trachea. We hypothesized that TLR ligands induce innate antiviral responses in the chicken trachea. Tracheal organ cultures (TOC) were used to investigate localized innate responses to TLR ligands. Expression of candidate genes, which play a role in antiviral responses, was quantified. To confirm the antiviral responses of stimulated TOC, chicken macrophages were treated with supernatants from stimulated TOC, prior to infection with AIV. The results demonstrated that TLR ligands induced the expression of pro-inflammatory cytokines, type I interferons and interferon stimulated genes in the chicken trachea. In conclusion, TLR ligands induce functional antiviral responses in the chicken trachea, which may act against some pathogens, such as AIV.
Keywords: TLR ligands; chicken trachea; innate antiviral responses.
Figures

Similar articles
-
Reduction of avian influenza virus shedding by administration of Toll-like receptor ligands to chickens.Vaccine. 2015 Sep 11;33(38):4843-9. doi: 10.1016/j.vaccine.2015.07.070. Epub 2015 Aug 1. Vaccine. 2015. PMID: 26238721
-
Innate antiviral responses are induced by TLR3 and TLR4 ligands in chicken tracheal epithelial cells: Communication between epithelial cells and macrophages.Virology. 2019 Aug;534:132-142. doi: 10.1016/j.virol.2019.06.003. Epub 2019 Jun 8. Virology. 2019. PMID: 31255797
-
Induction of antiviral responses against avian influenza virus in embryonated chicken eggs with toll-like receptor ligands.Viral Immunol. 2015 May;28(4):192-200. doi: 10.1089/vim.2014.0145. Epub 2015 Apr 15. Viral Immunol. 2015. PMID: 25874816
-
Immunostimulatory properties of Toll-like receptor ligands in chickens.Vet Immunol Immunopathol. 2013 Apr 15;152(3-4):191-9. doi: 10.1016/j.vetimm.2012.10.013. Epub 2012 Dec 13. Vet Immunol Immunopathol. 2013. PMID: 23305711 Review.
-
Contribution of Toll-like receptors to the control of hepatitis B virus infection by initiating antiviral innate responses and promoting specific adaptive immune responses.Cell Mol Immunol. 2015 May;12(3):273-82. doi: 10.1038/cmi.2014.112. Epub 2014 Nov 24. Cell Mol Immunol. 2015. PMID: 25418467 Free PMC article. Review.
Cited by
-
Distinct miRNA Profile of Cellular and Extracellular Vesicles Released from Chicken Tracheal Cells Following Avian Influenza Virus Infection.Vaccines (Basel). 2020 Aug 5;8(3):438. doi: 10.3390/vaccines8030438. Vaccines (Basel). 2020. PMID: 32764349 Free PMC article.
-
Characterization of the Role of Extracellular Vesicles Released from Chicken Tracheal Cells in the Antiviral Responses against Avian Influenza Virus.Membranes (Basel). 2021 Dec 31;12(1):53. doi: 10.3390/membranes12010053. Membranes (Basel). 2021. PMID: 35054579 Free PMC article.
-
Induction of Avian β-Defensin 2 Is Possibly Mediated by the p38 MAPK Signal Pathway in Chicken Embryo Fibroblasts After Newcastle Disease Virus Infection.Front Microbiol. 2018 Apr 19;9:751. doi: 10.3389/fmicb.2018.00751. eCollection 2018. Front Microbiol. 2018. PMID: 29725321 Free PMC article.
-
Primary cell culture systems to investigate host-pathogen interactions in bacterial respiratory tract infections of livestock.Front Cell Infect Microbiol. 2025 May 9;15:1565513. doi: 10.3389/fcimb.2025.1565513. eCollection 2025. Front Cell Infect Microbiol. 2025. PMID: 40415959 Free PMC article. Review.
-
Antiviral responses against chicken respiratory infections: Focus on avian influenza virus and infectious bronchitis virus.Cytokine. 2020 Mar;127:154961. doi: 10.1016/j.cyto.2019.154961. Epub 2019 Dec 31. Cytokine. 2020. PMID: 31901597 Free PMC article. Review.
References
-
- Reemers S.S., Groot Koerkamp M.J., Holstege F.C., van Eden W., Vervelde L. Cellular host transcriptional responses to influenza A virus in chicken tracheal organ cultures differ from responses in in vivo infected trachea. Vet. Immunol. Immunopathol. 2009;132:91–100. doi: 10.1016/j.vetimm.2009.04.021. - DOI - PubMed
-
- Matthijs M.G.R., Ariaans M.P., Dwars R.M., van Eck J.H.H., Bouma A., Stegeman A., Vervelde L., Van Eck J.H.H., Bouma A., Stegeman A., et al. Course of infection and immune responses in the respiratory tract of IBV infected broilers after superinfection with E. coli. Vet. Immunol. Immunopathol. 2009;127:77–84. doi: 10.1016/j.vetimm.2008.09.016. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

