Using CRISPR-Cas9 to quantify the contributions of O-glycans, N-glycans and Glycosphingolipids to human leukocyte-endothelium adhesion
- PMID: 27458028
- PMCID: PMC4960646
- DOI: 10.1038/srep30392
Using CRISPR-Cas9 to quantify the contributions of O-glycans, N-glycans and Glycosphingolipids to human leukocyte-endothelium adhesion
Abstract
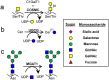
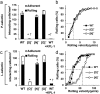
There is often interest in dissecting the relative contributions of the N-glycans, O-glycans and glycosphingolipids (GSLs) in regulating complex biological traits like cell signaling, adhesion, development and metastasis. To address this, we developed a CRISPR-Cas9 toolkit to selectively truncate each of these commonly expressed glycan-types. Here, O-glycan biosynthesis was truncated by knocking-out Core 1 β3Gal-T Specific Molecular Chaperone (COSMC), N-glycans by targeting the β1,2 GlcNAc-transferase (MGAT1) and GSLs by deleting UDP-glucose ceramide glucosyltransferase (UGCG). These reagents were applied to reveal the glycoconjugates regulating human myeloid cell adhesion to selectins under physiological shear-flow observed during inflammation. These functional studies show that leukocyte rolling on P- and L-selectin is ablated in cells lacking O-glycans, with N-glycan truncation also increasing cell rolling velocity on L-selectin. All three glycan families contributed to E-selectin dependent cell adhesion with N-glycans contributing to all aspects of the leukocyte adhesion cascade, O-glycans only being important during initial recruitment, and GSLs stabilizing slow cell rolling and the transition to firm arrest. Overall, the genome editing tools developed here may be broadly applied in studies of cellular glycosylation.
Figures





References
-
- Boscher C., Dennis J. W. & Nabi I. R. Glycosylation, galectins and cellular signaling. Curr Opin Cell Biol 23, 383–392 (2011). - PubMed
-
- Wright R. D. & Cooper D. Glycobiology of leukocyte trafficking in inflammation. Glycobiology 24, 1242–1251 (2014). - PubMed
-
- Varki A., Esko J. D. & Colley K. J. In Essentials of Glycobiology (eds Varki A. et al.) (2009).
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

