Structural role of the T94I rhodopsin mutation in congenital stationary night blindness
- PMID: 27458239
- PMCID: PMC5048376
- DOI: 10.15252/embr.201642671
Structural role of the T94I rhodopsin mutation in congenital stationary night blindness
Abstract
Congenital stationary night blindness (CSNB) is an inherited and non-progressive retinal dysfunction. Here, we present the crystal structure of CSNB-causing T94I2.61 rhodopsin in the active conformation at 2.3 Å resolution. The introduced hydrophobic side chain prolongs the lifetime of the G protein activating metarhodopsin-II state by establishing a direct van der Waals contact with K2967.43, the site of retinal attachment. This is in stark contrast to the light-activated state of the CSNB-causing G90D2.57 mutation, where the charged mutation forms a salt bridge with K2967.43 To find the common denominator between these two functional modifications, we combined our structural data with a kinetic biochemical analysis and molecular dynamics simulations. Our results indicate that both the charged G90D2.57 and the hydrophobic T94I2.61 mutation alter the dark state by weakening the interaction between the Schiff base (SB) and its counterion E1133.28 We propose that this interference with the tight regulation of the dim light photoreceptor rhodopsin increases background noise in the visual system and causes the loss of night vision characteristic for CSNB patients.
Keywords: G protein‐coupled receptors; congenital stationary night blindness; constitutive activity; rhodopsin; visual system.
© 2016 The Authors.
Figures

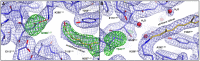
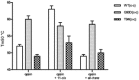

- A–C
Thermal decay of dark state rhodopsin: WT (A), T94I (B), and G90D (C) rhodopsin. The spectra are normalized to the OD280 and OD500/OD480 at t = 0.
- D
Absorption maximum plotted as a function of time and fitted to a single exponential decay.
- E–G
Retinal isomerization in the rhodopsin dark state: WT (E), T94I (F), and G90D (G) rhodopsin.
- H
The fraction of 11‐cis retinal is plotted as a function of time and fitted to a single exponential decay.
- I–K
SB stability in dark state rhodopsin: WT (I), T94I (J), and G90D (K) rhodopsin.
- L
OD440 is plotted as a function of time and fitted to a single exponential decay.
Similar articles
-
Insights into congenital stationary night blindness based on the structure of G90D rhodopsin.EMBO Rep. 2013 Jun;14(6):520-6. doi: 10.1038/embor.2013.44. Epub 2013 Apr 12. EMBO Rep. 2013. PMID: 23579341 Free PMC article.
-
Dark continuous noise from mutant G90D-rhodopsin predominantly underlies congenital stationary night blindness.Proc Natl Acad Sci U S A. 2024 May 21;121(21):e2404763121. doi: 10.1073/pnas.2404763121. Epub 2024 May 14. Proc Natl Acad Sci U S A. 2024. PMID: 38743626 Free PMC article.
-
Structural, energetic, and mechanical perturbations in rhodopsin mutant that causes congenital stationary night blindness.J Biol Chem. 2012 Jun 22;287(26):21826-35. doi: 10.1074/jbc.M112.340182. Epub 2012 May 1. J Biol Chem. 2012. PMID: 22549882 Free PMC article.
-
1 rhodopsin mutations in congenital night blindness.Adv Exp Med Biol. 2010;664:263-72. doi: 10.1007/978-1-4419-1399-9_30. Adv Exp Med Biol. 2010. PMID: 20238025 Review.
-
Structural aspects of rod opsin and their implication in genetic diseases.Pflugers Arch. 2021 Sep;473(9):1339-1359. doi: 10.1007/s00424-021-02546-x. Epub 2021 Mar 16. Pflugers Arch. 2021. PMID: 33728518 Review.
Cited by
-
G protein-coupled receptors: the evolution of structural insight.AIMS Biophys. 2017;4(3):491-527. doi: 10.3934/biophy.2017.3.491. Epub 2017 Aug 21. AIMS Biophys. 2017. PMID: 29951585 Free PMC article.
-
In Silico Studies Targeting G-protein Coupled Receptors for Drug Research Against Parkinson's Disease.Curr Neuropharmacol. 2018;16(6):786-848. doi: 10.2174/1570159X16666180308161642. Curr Neuropharmacol. 2018. PMID: 29521236 Free PMC article.
-
Stationary and Progressive Phenotypes Caused by the p.G90D Mutation in Rhodopsin Gene.Int J Mol Sci. 2021 Feb 21;22(4):2133. doi: 10.3390/ijms22042133. Int J Mol Sci. 2021. PMID: 33669941 Free PMC article.
-
A Conserved Proline Hinge Mediates Helix Dynamics and Activation of Rhodopsin.Structure. 2020 Sep 1;28(9):1004-1013.e4. doi: 10.1016/j.str.2020.05.004. Epub 2020 May 28. Structure. 2020. PMID: 32470317 Free PMC article.
-
Ligand channel in pharmacologically stabilized rhodopsin.Proc Natl Acad Sci U S A. 2018 Apr 3;115(14):3640-3645. doi: 10.1073/pnas.1718084115. Epub 2018 Mar 19. Proc Natl Acad Sci U S A. 2018. PMID: 29555765 Free PMC article.
References
-
- Smit MJ, Vischer HF, Bakker RA, Jongejan A, Timmerman H, Pardo L, Leurs R (2007) Pharmacogenomic and structural analysis of constitutive g protein‐coupled receptor activity. Annu Rev Pharmacol Toxicol 47: 53–87 - PubMed
-
- Robinson PRP, Cohen GBG, Zhukovsky EAE, Oprian DDD (1992) Constitutively active mutants of rhodopsin. Neuron 9: 719–725 - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

