Cellular Cations Control Conformational Switching of Inositol Pyrophosphate Analogues
- PMID: 27460418
- PMCID: PMC5076471
- DOI: 10.1002/chem.201601754
Cellular Cations Control Conformational Switching of Inositol Pyrophosphate Analogues
Abstract
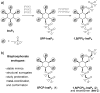
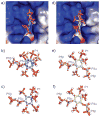
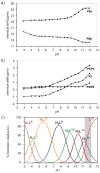
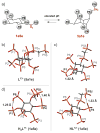
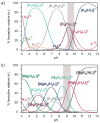
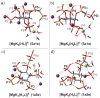
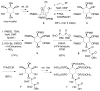
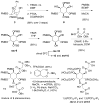
The inositol pyrophosphate messengers (PP-InsPs) are emerging as an important class of cellular regulators. These molecules have been linked to numerous biological processes, including insulin secretion and cancer cell migration, but how they trigger such a wide range of cellular responses has remained unanswered in many cases. Here, we show that the PP-InsPs exhibit complex speciation behaviour and propose that a unique conformational switching mechanism could contribute to their multifunctional effects. We synthesised non-hydrolysable bisphosphonate analogues and crystallised the analogues in complex with mammalian PPIP5K2 kinase. Subsequently, the bisphosphonate analogues were used to investigate the protonation sequence, metal-coordination properties, and conformation in solution. Remarkably, the presence of potassium and magnesium ions enabled the analogues to adopt two different conformations near physiological pH. Understanding how the intrinsic chemical properties of the PP-InsPs can contribute to their complex signalling outputs will be essential to elucidate their regulatory functions.
Keywords: biological activity; conformation analysis; phosphorylation; protonation; signal transduction.
© 2016 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures

References
-
- Shears SB. Adv Biol Regul. 2015;57:203–216. - PMC - PubMed
- Wilson MS, Livermore TM, Saiardi A. Biochem J. 2013;452:369–379. - PubMed
- Wundenberg T, Mayr GW. Biol Chem. 2012;393:979–998. - PubMed
- Chakraborty A, Kim S, Snyder SH. Science Signal. 2011;4:re1–11. - PMC - PubMed
- Barker CJ, Illies C, Gaboardi GC, Berggren PO. Cell Mol Life Sci. 2009;66:3851–3871. - PMC - PubMed
- Monserrate JP, York JD. Curr Opin Cell Biol. 2010;22:365–373. - PubMed
-
- Saiardi A, Erdjument-Bromage H, Snowman AM, Tempst P, Snyder SH. Curr Biol. 1999;9:1323–1326. - PubMed
- Saiardi A, Nagata E, Luo HR, Snowman AM, Snyder SH. J Biol Chem. 2001;276:39179–39185. - PubMed
- Schell MJ, Letcher AJ, Brearley CA, Biber J, Murer H, Irvine RF. FEBS Lett. 1999;461:169–172. - PubMed
- Voglmaier SM, Bembenek ME, Kaplin AI, Dorman G, Olszewski JD, Prestwich GD, Snyder SH. Proc Natl Acad Sci USA. 1996;93:4305–4310. - PMC - PubMed
-
- Mulugu S, Bai W, Fridy PC, Bastidas RJ, Otto JC, Dollins DE, Haystead TA, Ribeiro AA, York JD. Science. 2007;316:106–109. - PubMed
- Choi JH, Williams J, Cho J, Falck JR, Shears SB. J Biol Chem. 2007;282:30763–30775. - PMC - PubMed
- Lin H, Fridy PC, Ribeiro AA, Choi JH, Barma DK, Vogel G, Falck JR, Shears SB, York JD, Mayr GW. J Biol Chem. 2008;284:1863–1872. - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

