Assessment of whole blood thrombosis in a microfluidic device lined by fixed human endothelium
- PMID: 27464497
- PMCID: PMC4963439
- DOI: 10.1007/s10544-016-0095-6
Assessment of whole blood thrombosis in a microfluidic device lined by fixed human endothelium
Abstract
The vascular endothelium and shear stress are critical determinants of physiological hemostasis and platelet function in vivo, yet current diagnostic and monitoring devices do not fully incorporate endothelial function under flow in their assessment and, therefore, they can be unreliable and inaccurate. It is challenging to include the endothelium in assays for clinical laboratories or point-of-care settings because living cell cultures are not sufficiently robust. Here, we describe a microfluidic device that is lined by a human endothelium that is chemically fixed, but still retains its ability to modulate hemostasis under continuous flow in vitro even after few days of storage. This device lined with a fixed endothelium supports formation of platelet-rich thrombi in the presence of physiological shear, similar to a living arterial vessel. We demonstrate the potential clinical value of this device by showing that thrombus formation and platelet function can be measured within minutes using a small volume (0.5 mL) of whole blood taken from subjects receiving antiplatelet medications. The inclusion of a fixed endothelial microvessel will lead to biomimetic analytical devices that can potentially be used for diagnostics and point-of-care applications.
Keywords: Biomedical technology; Hemostasis; Lab-on-a-Chip; Platelet function tests; Thrombosis; Vascular endothelium.
Figures
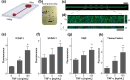
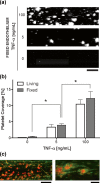
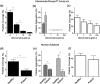
 or collagen
or collagen  as an agonist (n = 4). c Platelet coverage when blood samples containing different doses of the drug abciximab were perfused through collagen-coated microfluidic devices (n = 4). d Platelet coverage on the fixed endothelium pretreated with TNF-α when blood samples from healthy donors versus subjects treated with antiplatelet drugs were perfused through microfluidic devices (n = 11). e Light transmission aggregometry of healthy versus antiplatelet treated blood samples using ADP
as an agonist (n = 4). c Platelet coverage when blood samples containing different doses of the drug abciximab were perfused through collagen-coated microfluidic devices (n = 4). d Platelet coverage on the fixed endothelium pretreated with TNF-α when blood samples from healthy donors versus subjects treated with antiplatelet drugs were perfused through microfluidic devices (n = 11). e Light transmission aggregometry of healthy versus antiplatelet treated blood samples using ADP  or collagen
or collagen  as an agonist (n = 11). f Platelet coverage when healthy versus subject blood samples were perfused through collagen-coated microfluidic devices (n = 11). *P < 0.05
as an agonist (n = 11). f Platelet coverage when healthy versus subject blood samples were perfused through collagen-coated microfluidic devices (n = 11). *P < 0.05References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

