Effect of gadolinium-based nanoparticles on nuclear DNA damage and repair in glioblastoma tumor cells
- PMID: 27464501
- PMCID: PMC4964094
- DOI: 10.1186/s12951-016-0215-8
Effect of gadolinium-based nanoparticles on nuclear DNA damage and repair in glioblastoma tumor cells
Abstract
Background: Tumor targeting of radiotherapy represents a great challenge. The addition of multimodal nanoparticles, such as 3 nm gadolinium-based nanoparticles (GdBNs), has been proposed as a promising strategy to amplify the effects of radiation in tumors and improve diagnostics using the same agents. This singular property named theranostic is a unique advantage of GdBNs. It has been established that the amplification of radiation effects by GdBNs appears due to fast electronic processes. However, the influence of these nanoparticles on cells is not yet understood. In particular, it remains dubious how nanoparticles activated by ionizing radiation interact with cells and their constituents. A crucial question remains open of whether damage to the nucleus is necessary for the radiosensitization exerted by GdBNs (and other nanoparticles).
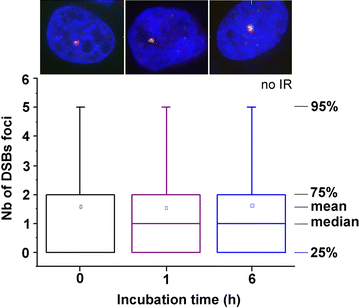
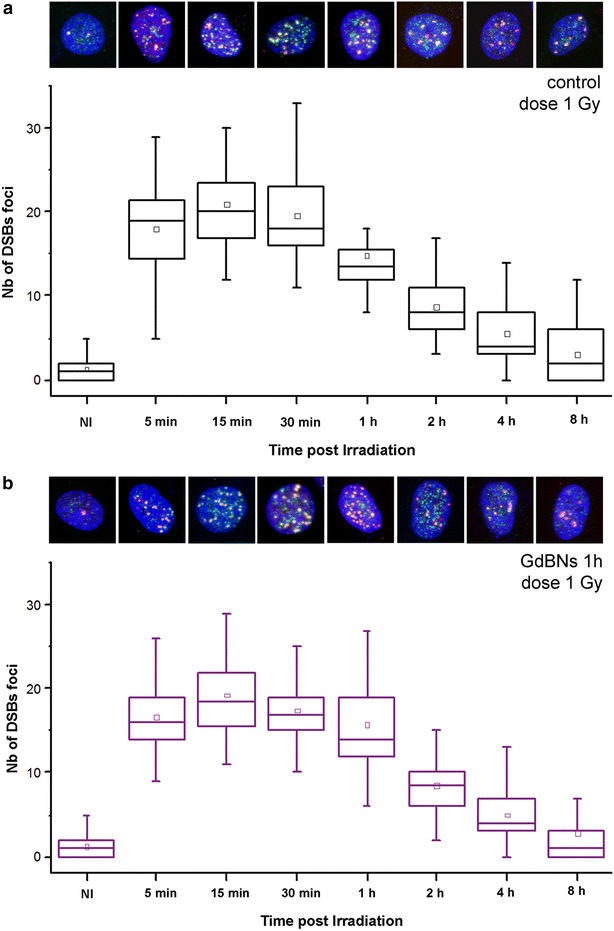
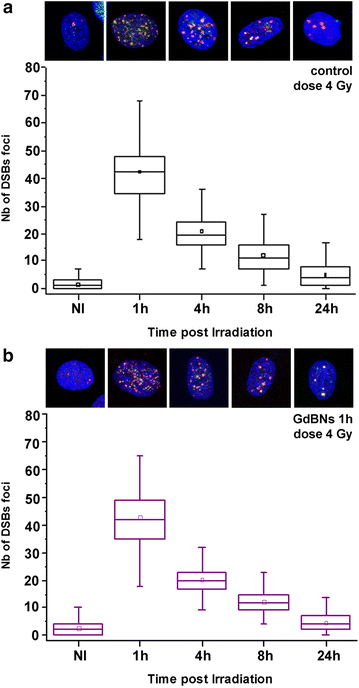
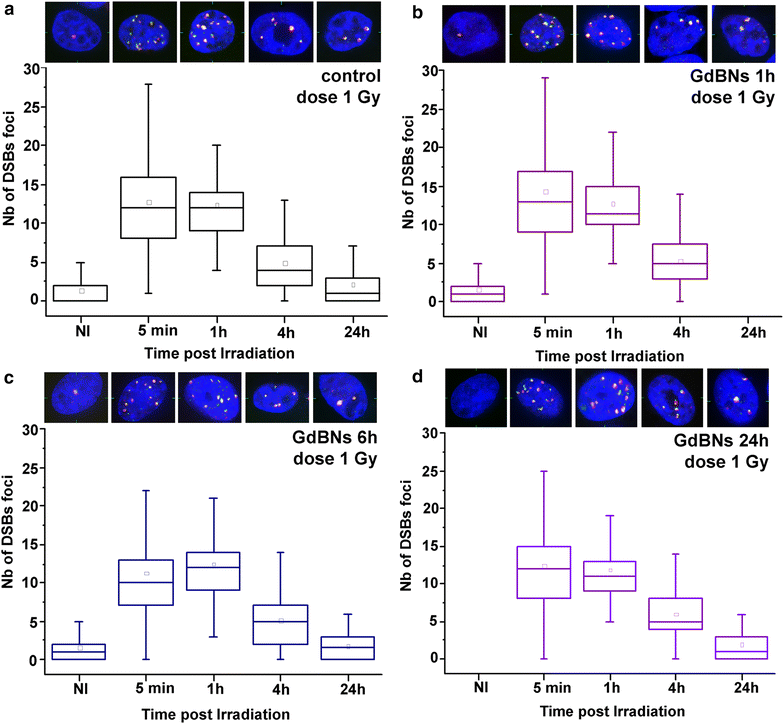
Methods: We studied the effect of GdBNs on the induction and repair of DNA double-strand breaks (DSBs) in the nuclear DNA of U87 tumor cells irradiated with γ-rays. For this purpose, we used currently the most sensitive method of DSBs detection based on high-resolution confocal fluorescence microscopy coupled with immunodetection of two independent DSBs markers.
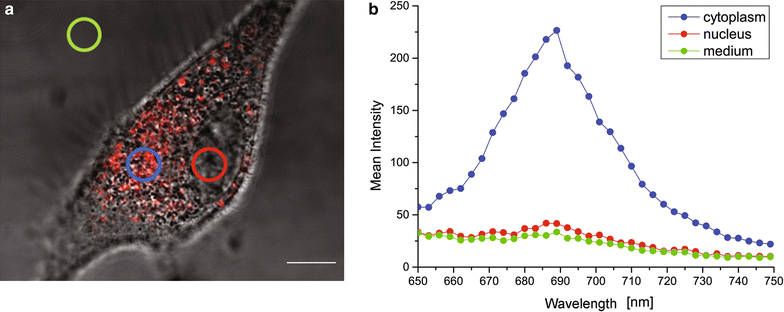
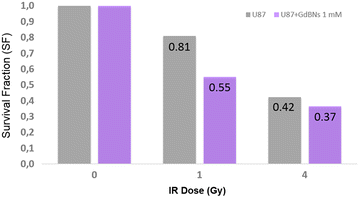
Results: We show that, in the conditions where GdBNs amplify radiation effects, they remain localized in the cytoplasm, i.e. do not penetrate into the nucleus. In addition, the presence of GdBNs in the cytoplasm neither increases induction of DSBs by γ-rays in the nuclear DNA nor affects their consequent repair.
Conclusions: Our results suggest that the radiosensitization mediated by GdBNs is a cytoplasmic event that is independent of the nuclear DNA breakage, a phenomenon commonly accepted as the explanation of biological radiation effects. Considering our earlier recognized colocalization of GdBNs with the lysosomes and endosomes, we revolutionary hypothesize here about these organelles as potential targets for (some) nanoparticles. If confirmed, this finding of cytoplasmically determined radiosensitization opens new perspectives of using nano-radioenhancers to improve radiotherapy without escalating the risk of pathologies related to genetic damage.
Keywords: DNA double-strand breaks; DNA repair; Gadolinium; Nanomedicine; Nanoparticles; Radiosensitization; Radiotherapy; Theranostic.
Figures

Similar articles
-
Challenges and Contradictions of Metal Nano-Particle Applications for Radio-Sensitivity Enhancement in Cancer Therapy.Int J Mol Sci. 2019 Jan 30;20(3):588. doi: 10.3390/ijms20030588. Int J Mol Sci. 2019. PMID: 30704035 Free PMC article.
-
Radiosensitization Effect of AGuIX, a Gadolinium-Based Nanoparticle, in Nonsmall Cell Lung Cancer.ACS Appl Mater Interfaces. 2020 Dec 23;12(51):56874-56885. doi: 10.1021/acsami.0c16548. Epub 2020 Dec 16. ACS Appl Mater Interfaces. 2020. PMID: 33326207
-
Mechanism of radiosensitization by halogenated pyrimidines: effect of BrdU on repair of DNA breaks, interphase chromatin breaks, and potentially lethal damage in plateau-phase CHO cells.Radiat Res. 1992 Feb;129(2):202-11. Radiat Res. 1992. PMID: 1734451
-
DNA double strand break repair inhibition as a cause of heat radiosensitization: re-evaluation considering backup pathways of NHEJ.Int J Hyperthermia. 2008 Feb;24(1):17-29. doi: 10.1080/02656730701784782. Int J Hyperthermia. 2008. PMID: 18214766 Review.
-
Mechanisms of DNA double strand break repair and chromosome aberration formation.Cytogenet Genome Res. 2004;104(1-4):14-20. doi: 10.1159/000077461. Cytogenet Genome Res. 2004. PMID: 15162010 Review.
Cited by
-
Radiosensitizing high-Z metal nanoparticles for enhanced radiotherapy of glioblastoma multiforme.J Nanobiotechnology. 2020 Sep 3;18(1):122. doi: 10.1186/s12951-020-00684-5. J Nanobiotechnology. 2020. PMID: 32883290 Free PMC article. Review.
-
Proof of Concept of the Radiosensitizing Effect of Gadolinium Oxide Nanoparticles in Cell Spheroids and a Tumor-Implanted Murine Model of Chondrosarcoma.Int J Nanomedicine. 2022 Dec 23;17:6655-6673. doi: 10.2147/IJN.S390056. eCollection 2022. Int J Nanomedicine. 2022. PMID: 36582458 Free PMC article. Clinical Trial.
-
Combinational Radiotherapies Improve Brain Cancer Treatment at High Dose Rates In Vitro.Cancers (Basel). 2025 May 20;17(10):1713. doi: 10.3390/cancers17101713. Cancers (Basel). 2025. PMID: 40427210 Free PMC article.
-
Prospects of nanoparticle-based radioenhancement for radiotherapy.Mater Horiz. 2023 Oct 2;10(10):4059-4082. doi: 10.1039/d3mh00265a. Mater Horiz. 2023. PMID: 37555747 Free PMC article. Review.
-
Targeting brain metastases with ultrasmall theranostic nanoparticles, a first-in-human trial from an MRI perspective.Sci Adv. 2020 Jul 15;6(29):eaay5279. doi: 10.1126/sciadv.aay5279. eCollection 2020 Jul. Sci Adv. 2020. PMID: 32832613 Free PMC article.
References
-
- Matsumura Y, Maeda H. A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986;46:6387–6392. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

