Clinical-Grade Human Multipotent Adult Progenitor Cells Block CD8+ Cytotoxic T Lymphocytes
- PMID: 27465071
- PMCID: PMC5189653
- DOI: 10.5966/sctm.2016-0030
Clinical-Grade Human Multipotent Adult Progenitor Cells Block CD8+ Cytotoxic T Lymphocytes
Abstract
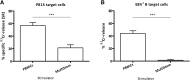
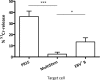
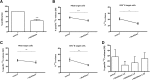
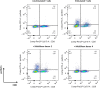
: MultiStem cells are clinical-grade multipotent adult bone marrow-derived progenitor cells (MAPCs), with extensive replication potential and broader differentiation capacity compared with mesenchymal stem cells. Human MAPCs suppress T-cell proliferation induced by alloantigens and mutually interact with allogeneic natural killer cells. In this study, the interaction between MultiStem and CD8+ cytotoxic T lymphocytes (CTLs) was addressed for the first time. In an in vitro setting, the immunogenicity of MultiStem, the susceptibility of MultiStem toward CTL-mediated lysis, and its effects on CTL function were investigated. MultiStem was nonimmunogenic for alloreactive CTL induction and was-even after major histocompatibility complex class I upregulation-insensitive to alloantigen-specific CTL-mediated lysis. Furthermore, MultiStem reduced CTL proliferation and significantly decreased perforin expression during the T-cell activation phase. As a consequence, MultiStem dose-dependently impaired the induction of CTL function. These effects of MultiStem were mediated predominantly through contact-dependent mechanisms. Moreover, MultiStem cells considerably influenced the expression of T-cell activation markers CD25, CD69, and human leukocyte antigen-DR. The MultiStem-induced CD8-CD69+ T-cell population displayed a suppressive effect on the induction of CTL function during a subsequent mixed-lymphocyte culture. Finally, the killer activity of activated antigen-specific CTLs during their cytolytic effector phase was also diminished in the presence of MultiStem. This study confirms that these clinical-grade MAPCs are an immune-modulating population that inhibits CTL activation and effector responses and are, consequently, a highly valuable cell population for adoptive immunosuppressive therapy in diseases where damage is induced by CTLs.
Significance: Because multipotent adult progenitor cells (MAPCs) are among the noteworthy adult mesenchymal stem cell populations for immune therapy and have the advantage over mesenchymal stem cells (MSCs) of large-scale manufacturing and banking potential and thus prompt availability, it is important to understand how MAPCs interact with immune cells to validate their widespread therapeutic applicability. Cytotoxic immune effector cells play a crucial role in immune homeostasis and in the pathogenesis of some autoimmune diseases. This study assessed for the first time the in vitro influence of a clinical-grade human MAPC product (MultiStem) on the cytotoxic function of CD8+ T cells (CTLs) by evaluating the immunogenicity of MAPCs and the susceptibility of MAPCs toward CTL-mediated lysis and by analyzing the mechanism of MAPC-mediated modulation of CTL functionality. These results may represent a highly relevant contribution to the current knowledge and, in combination with the results of future phase II/III trials using MultiStem, could lead to an intriguing continuation of stem cell-based research for immunotherapy.
Keywords: Clinical-grade human multipotent adult progenitor cells; Cytotoxic T cells; MultiStem; Stem cell-based immune modulation; T cell-mediated cytotoxicity.
©AlphaMed Press.
Figures



Similar articles
-
Solution-Phase Crosstalk and Regulatory Interactions Between Multipotent Adult Progenitor Cells and Peripheral Blood Mononuclear Cells.Stem Cells Transl Med. 2015 Dec;4(12):1436-49. doi: 10.5966/sctm.2014-0225. Epub 2015 Oct 22. Stem Cells Transl Med. 2015. PMID: 26494783 Free PMC article.
-
Human multipotent adult progenitor cells are nonimmunogenic and exert potent immunomodulatory effects on alloreactive T-cell responses.Cell Transplant. 2013;22(10):1915-28. doi: 10.3727/096368912X657369. Epub 2012 Oct 1. Cell Transplant. 2013. PMID: 23031260
-
Mutual interaction between human multipotent adult progenitor cells and NK cells.Cell Transplant. 2014;23(9):1099-110. doi: 10.3727/096368913X665585. Cell Transplant. 2014. PMID: 23562064
-
Cellular mechanisms of lymphocyte-mediated lysis of tumor cells.Ann Ist Super Sanita. 1990;26(3-4):369-84. Ann Ist Super Sanita. 1990. PMID: 2151107 Review.
-
A comparison of the cytolytic properties of murine primary CD8+ cytotoxic T lymphocytes and cloned cytotoxic T cell lines.Immunol Rev. 1988 Mar;103:111-25. doi: 10.1111/j.1600-065x.1988.tb00753.x. Immunol Rev. 1988. PMID: 3134291 Review.
Cited by
-
Multipotent adult progenitor cells induce regulatory T cells and promote their suppressive phenotype via TGFβ and monocyte-dependent mechanisms.Sci Rep. 2021 Jun 30;11(1):13549. doi: 10.1038/s41598-021-93025-x. Sci Rep. 2021. PMID: 34193955 Free PMC article.
-
The Delivery of Multipotent Adult Progenitor Cells to Extended Criteria Human Donor Livers Using Normothermic Machine Perfusion.Front Immunol. 2020 Jun 25;11:1226. doi: 10.3389/fimmu.2020.01226. eCollection 2020. Front Immunol. 2020. PMID: 32714318 Free PMC article.
-
Pluripotent Stem Cells in Adult Tissues: Struggling To Be Acknowledged Over Two Decades.Stem Cell Rev Rep. 2017 Dec;13(6):713-724. doi: 10.1007/s12015-017-9756-y. Stem Cell Rev Rep. 2017. PMID: 28815441 Review.
-
Molecular Classification and Tumor Microenvironment Characterization of Gallbladder Cancer by Comprehensive Genomic and Transcriptomic Analysis.Cancers (Basel). 2021 Feb 10;13(4):733. doi: 10.3390/cancers13040733. Cancers (Basel). 2021. PMID: 33578820 Free PMC article.
-
Alginate encapsulated multipotent adult progenitor cells promote corneal stromal cell activation via release of soluble factors.PLoS One. 2018 Sep 7;13(9):e0202118. doi: 10.1371/journal.pone.0202118. eCollection 2018. PLoS One. 2018. PMID: 30192833 Free PMC article.
References
-
- Bongso A, Lee EH. Stem cells: Their definition, classification and sources. In: Bongso A, Lee EH, eds. Stem Cells: From Bench to Bedside. Toh Tuck Link, Singapore: World Scientific Publishing, 2005:1–13
-
- Friedenstein AJ, Deriglasova UF, Kulagina NN, et al. Precursors for fibroblasts in different populations of hematopoietic cells as detected by the in vitro colony assay method. Exp Hematol. 1974;2:83–92. - PubMed
-
- Prockop DJ. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science. 1997;276:71–74. - PubMed
-
- Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. - PubMed
-
- da Silva Meirelles L, Chagastelles PC, Nardi NB. Mesenchymal stem cells reside in virtually all post-natal organs and tissues. J Cell Sci. 2006;119:2204–2213. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

