Primacy of cardiac chymase over angiotensin converting enzyme as an angiotensin-(1-12) metabolizing enzyme
- PMID: 27465904
- PMCID: PMC5207032
- DOI: 10.1016/j.bbrc.2016.07.100
Primacy of cardiac chymase over angiotensin converting enzyme as an angiotensin-(1-12) metabolizing enzyme
Abstract
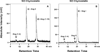
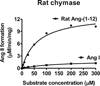
We showed previously that rat angiotensin-(1-12) [Ang-(1-12)] is metabolized by chymase and angiotensin converting enzyme (ACE) to generate Angiotensin II (Ang II). Here, we investigated the affinity of cardiac chymase and ACE enzymes for Ang-(1-12) and Angiotensin I (Ang I) substrates. Native plasma membranes (PMs) isolated from heart and lung tissues of adult spontaneously hypertensive rats (SHR) were incubated with radiolabeled (125)I-Ang-(1-12) or (125)I-Ang I, in the absence or presence of a chymase or ACE inhibitor (chymostatin and lisinopril, respectively). Products were quantitated by HPLC connected to an in-line flow-through gamma detector. The rate of (125)I-Ang II formation from (125)I-Ang-(1-12) by chymase was significantly higher (heart: 7.0 ± 0.6 fmol/min/mg; lung: 33 ± 1.2 fmol/min/mg, P < 0.001) when compared to (125)I-Ang I substrate (heart: 0.8 ± 0.1 fmol/min/mg; lung: 2.1 ± 0.1 fmol/min/mg). Substrate affinity of (125)I-Ang-(1-12) for rat cardiac chymase was also confirmed using excess unlabeled Ang-(1-12) or Ang I (0-250 μM). The rate of (125)I-Ang II formation was significantly lower using unlabeled Ang-(1-12) compared to unlabeled Ang I substrate. Kinetic data showed that rat chymase has a lower Km (64 ± 6.3 μM vs 142 ± 17 μM), higher Vmax (13.2 ± 1.3 μM/min/mg vs 1.9 ± 0.2 μM/min/mg) and more than 15-fold higher catalytic efficiency (ratio of Vmax/Km) for Ang-(1-12) compared to Ang I substrate, respectively. We also investigated ACE mediated hydrolysis of (125)I-Ang-(1-12) and (125)I-Ang I in solubilized membrane fractions of the SHR heart and lung. Interestingly, no significant difference in (125)I-Ang II formation by ACE was detected using either substrate, (125)I-Ang-(1-12) or (125)I-Ang I, both in the heart (1.8 ± 0.2 fmol/min/mg and 1.8 ± 0.3 fmol/min/mg, respectively) and in the lungs (239 ± 25 fmol/min/mg and 248 ± 34 fmol/min/mg, respectively). Compared to chymase, ACE-mediated Ang-(1-12) metabolism in the heart was several fold lower. Overall our findings suggest that Ang-(1-12), not Ang I, is the better substrate for Ang II formation by chymase in adult rats. In addition, this confirms our previous observation that chymase (rather than ACE) is the main hydrolyzing enzyme responsible for Ang II generation from Ang-(1-12) in the adult rat heart.
Keywords: Angiotensin I; Angiotensin II; Angiotensin-(1-12); Angiotensin-converting enzyme; Rat cardiac chymase; Renin-angiotensin system.
Copyright © 2016 Elsevier Inc. All rights reserved.
Conflict of interest statement
All authors have no potential conflicts of interest relevant to this article. Disclosure No potential conflicts of interest relevant to this article.
Figures


Similar articles
-
Chymase-dependent generation of angiotensin II from angiotensin-(1-12) in human atrial tissue.PLoS One. 2011;6(12):e28501. doi: 10.1371/journal.pone.0028501. Epub 2011 Dec 13. PLoS One. 2011. PMID: 22180785 Free PMC article.
-
Primacy of angiotensin converting enzyme in angiotensin-(1-12) metabolism.Am J Physiol Heart Circ Physiol. 2013 Sep 1;305(5):H644-50. doi: 10.1152/ajpheart.00210.2013. Epub 2013 Jun 28. Am J Physiol Heart Circ Physiol. 2013. PMID: 23812388 Free PMC article.
-
Chymase-dependent production of angiotensin II: an old enzyme in old hearts.Am J Physiol Heart Circ Physiol. 2017 Feb 1;312(2):H223-H231. doi: 10.1152/ajpheart.00534.2016. Epub 2016 Nov 4. Am J Physiol Heart Circ Physiol. 2017. PMID: 27815252 Free PMC article.
-
Angiotensin I converting enzyme and chymase in cardiovascular tissues.Kidney Int Suppl. 1996 Jun;55:S18-23. Kidney Int Suppl. 1996. PMID: 8743505 Review.
-
Novel Cardiac Intracrine Mechanisms Based on Ang-(1-12)/Chymase Axis Require a Revision of Therapeutic Approaches in Human Heart Disease.Curr Hypertens Rep. 2017 Feb;19(2):16. doi: 10.1007/s11906-017-0708-3. Curr Hypertens Rep. 2017. PMID: 28233239 Free PMC article. Review.
Cited by
-
Does the Naked Emperor Parable Apply to Current Perceptions of the Contribution of Renin Angiotensin System Inhibition in Hypertension?Curr Hypertens Rep. 2022 Dec;24(12):709-721. doi: 10.1007/s11906-022-01229-x. Epub 2022 Oct 22. Curr Hypertens Rep. 2022. PMID: 36272015 Review.
-
Chymase inhibitors for the treatment of cardiac diseases: a patent review (2010-2018).Expert Opin Ther Pat. 2018 Nov;28(11):755-764. doi: 10.1080/13543776.2018.1531848. Epub 2018 Oct 10. Expert Opin Ther Pat. 2018. PMID: 30278800 Free PMC article. Review.
-
Is chymase 1 a therapeutic target in cardiovascular disease?Expert Opin Ther Targets. 2023 Jul-Dec;27(8):645-656. doi: 10.1080/14728222.2023.2247561. Epub 2023 Aug 21. Expert Opin Ther Targets. 2023. PMID: 37565266 Free PMC article. Review.
-
Collecting Duct Nitric Oxide Synthase 1ß Activation Maintains Sodium Homeostasis During High Sodium Intake Through Suppression of Aldosterone and Renal Angiotensin II Pathways.J Am Heart Assoc. 2017 Oct 24;6(10):e006896. doi: 10.1161/JAHA.117.006896. J Am Heart Assoc. 2017. PMID: 29066445 Free PMC article.
-
Renin angiotensin aldosterone inhibition in the treatment of cardiovascular disease.Pharmacol Res. 2017 Nov;125(Pt A):57-71. doi: 10.1016/j.phrs.2017.05.020. Epub 2017 May 29. Pharmacol Res. 2017. PMID: 28571891 Free PMC article. Review.
References
-
- Ferrario CM, Chappell MC, Dean RH, Iyer SN. Novel angiotensin peptides regulate blood pressure, endothelial function, and natriuresis. J. Am. Soc. Nephrol. 1998;9:1716–1722. - PubMed
-
- Mendoza-Torres E, Oyarzun A, Mondaca-Ruff D, Azocar A, Cartro PF, Jalil JE, Chiong M, Lavandero S, Ocaranza MP. ACE2 and vasoactive peptides: novel players in cardiovascular/renal remodeling and hypertension. Ther. Adv. Cardiovasc. Dis. 2015;9:217–237. - PubMed
-
- Nagata S, Kato J, Sasaki K, Minamino N, Eto T, Kitamura K. Isolation and identification of proangiotensin-12, a possible component of the renin-angiotensin system. Biochem. Biophys. Res. Commun. 2006;350:1026–1031. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

