Neural Responses to Heartbeats in the Default Network Encode the Self in Spontaneous Thoughts
- PMID: 27466329
- PMCID: PMC4961773
- DOI: 10.1523/JNEUROSCI.0262-16.2016
Neural Responses to Heartbeats in the Default Network Encode the Self in Spontaneous Thoughts
Abstract
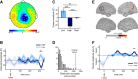
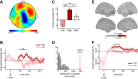
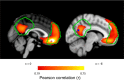
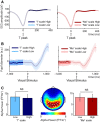
The default network (DN) has been consistently associated with self-related cognition, but also to bodily state monitoring and autonomic regulation. We hypothesized that these two seemingly disparate functional roles of the DN are functionally coupled, in line with theories proposing that selfhood is grounded in the neural monitoring of internal organs, such as the heart. We measured with magnetoencephalograhy neural responses evoked by heartbeats while human participants freely mind-wandered. When interrupted by a visual stimulus at random intervals, participants scored the self-relatedness of the interrupted thought. They evaluated their involvement as the first-person perspective subject or agent in the thought ("I"), and on another scale to what degree they were thinking about themselves ("Me"). During the interrupted thought, neural responses to heartbeats in two regions of the DN, the ventral precuneus and the ventromedial prefrontal cortex, covaried, respectively, with the "I" and the "Me" dimensions of the self, even at the single-trial level. No covariation between self-relatedness and peripheral autonomic measures (heart rate, heart rate variability, pupil diameter, electrodermal activity, respiration rate, and phase) or alpha power was observed. Our results reveal a direct link between selfhood and neural responses to heartbeats in the DN and thus directly support theories grounding selfhood in the neural monitoring of visceral inputs. More generally, the tight functional coupling between self-related processing and cardiac monitoring observed here implies that, even in the absence of measured changes in peripheral bodily measures, physiological and cognitive functions have to be considered jointly in the DN.
Significance statement: The default network (DN) has been consistently associated with self-processing but also with autonomic regulation. We hypothesized that these two functions could be functionally coupled in the DN, inspired by theories according to which selfhood is grounded in the neural monitoring of internal organs. Using magnetoencephalography, we show that heartbeat-evoked responses (HERs) in the DN covary with the self-relatedness of ongoing spontaneous thoughts. HER amplitude in the ventral precuneus covaried with the "I" self-dimension, whereas HER amplitude in the ventromedial prefrontal cortex encoded the "Me" self-dimension. Our experimental results directly support theories rooting selfhood in the neural monitoring of internal organs. We propose a novel functional framework for the DN, where self-processing is coupled with physiological monitoring.
Keywords: MEG; default network; heartbeat-evoked responses; self; spontaneous cognition.
Copyright © 2016 Babo-Rebelo et al.
Figures

Similar articles
-
Is the cardiac monitoring function related to the self in both the default network and right anterior insula?Philos Trans R Soc Lond B Biol Sci. 2016 Nov 19;371(1708):20160004. doi: 10.1098/rstb.2016.0004. Epub 2016 Oct 10. Philos Trans R Soc Lond B Biol Sci. 2016. PMID: 28080963 Free PMC article.
-
Responses to Heartbeats in Ventromedial Prefrontal Cortex Contribute to Subjective Preference-Based Decisions.J Neurosci. 2021 Jun 9;41(23):5102-5114. doi: 10.1523/JNEUROSCI.1932-20.2021. Epub 2021 Apr 29. J Neurosci. 2021. PMID: 33926998 Free PMC article.
-
Shaped by our thoughts--a new task to assess spontaneous cognition and its associated neural correlates in the default network.Brain Cogn. 2015 Feb;93:1-10. doi: 10.1016/j.bandc.2014.11.001. Epub 2014 Nov 18. Brain Cogn. 2015. PMID: 25463243
-
The neural monitoring of visceral inputs, rather than attention, accounts for first-person perspective in conscious vision.Cortex. 2018 May;102:139-149. doi: 10.1016/j.cortex.2017.05.019. Epub 2017 Jun 3. Cortex. 2018. PMID: 28651745 Review.
-
A framework for understanding the relationship between externally and internally directed cognition.Neuropsychologia. 2014 Sep;62:321-30. doi: 10.1016/j.neuropsychologia.2014.05.024. Epub 2014 Jun 6. Neuropsychologia. 2014. PMID: 24912071 Review.
Cited by
-
Affect and Social Judgment: The Roles of Physiological Reactivity and Interoceptive Sensitivity.Affect Sci. 2022 Apr 28;3(2):464-479. doi: 10.1007/s42761-022-00114-9. eCollection 2022 Jun. Affect Sci. 2022. PMID: 36046009 Free PMC article.
-
Attention to cardiac sensations enhances the heartbeat-evoked potential during exhalation.iScience. 2024 Mar 27;27(4):109586. doi: 10.1016/j.isci.2024.109586. eCollection 2024 Apr 19. iScience. 2024. PMID: 38623333 Free PMC article.
-
Altered processing of self-produced sensations in psychosis at cortical and spinal levels.Mol Psychiatry. 2025 Jul 25. doi: 10.1038/s41380-025-03130-w. Online ahead of print. Mol Psychiatry. 2025. PMID: 40715480
-
Meditation and interoception: a conceptual framework for the narrative and experiential self.Front Psychol. 2024 Oct 16;15:1393969. doi: 10.3389/fpsyg.2024.1393969. eCollection 2024. Front Psychol. 2024. PMID: 39478794 Free PMC article. Review.
-
Temporal Relations between Cortical Network Oscillations and Breathing Frequency during REM Sleep.J Neurosci. 2021 Jun 16;41(24):5229-5242. doi: 10.1523/JNEUROSCI.3067-20.2021. Epub 2021 May 7. J Neurosci. 2021. PMID: 33963051 Free PMC article.
References
-
- Armour JA, Ardell JL. Basic and clinical neurocardiology. In: Armour JA, Ardell JL, editors. New York: Oxford UP; 2004.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
