Modulation of Phosphopeptide Fragmentation via Dual Spray Ion/Ion Reactions Using a Sulfonate-Incorporating Reagent
- PMID: 27467576
- PMCID: PMC5477467
- DOI: 10.1021/acs.analchem.6b01901
Modulation of Phosphopeptide Fragmentation via Dual Spray Ion/Ion Reactions Using a Sulfonate-Incorporating Reagent
Abstract
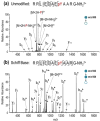
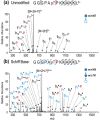
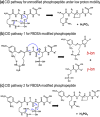
The labile nature of phosphoryl groups has presented a long-standing challenge for the characterization of protein phosphorylation via conventional mass spectrometry-based bottom-up proteomics methods. Collision-induced dissociation (CID) causes preferential cleavage of the phospho-ester bond of peptides, particularly under conditions of low proton mobility, and results in the suppression of sequence-informative fragmentation that often prohibits phosphosite determination. In the present study, the fragmentation patterns of phosphopeptides are improved through ion/ion-mediated peptide derivatization with 4-formyl-1,3-benezenedisulfonic acid (FBDSA) anions using a dual spray reactor. This approach exploits the strong electrostatic interactions between the sulfonate moieties of FBDSA and basic sites to facilitate gas-phase bioconjugation and to reduce charge sequestration and increase the yield of phosphate-retaining sequence ions upon CID. Moreover, comparative CID fragmentation analysis between unmodified phosphopeptides and those modified online with FBDSA or in solution via carbamylation and 4-sulfophenyl isothiocyanate (SPITC) provided evidence for sulfonate interference with charge-directed mechanisms that result in preferential phosphate elimination. Our results indicate the prominence of charge-directed neighboring group participation reactions involved in phosphate neutral loss, and the implementation of ion/ion reactions in a dual spray reactor setup provides a means to disrupt the interactions by competing hydrogen-bonding interactions between sulfonate groups and the side chains of basic residues.
Figures


Similar articles
-
Observation of an unusually facile fragmentation pathway of gas-phase peptide ions: a study on the gas-phase fragmentation mechanism and energetics of tryptic peptides modified with 4-sulfophenyl isothiocyanate (SPITC) and 4-chlorosulfophenyl isocyanate (SPC) and their 18-crown-6 complexes.J Mass Spectrom. 2007 Mar;42(3):380-8. doi: 10.1002/jms.1172. J Mass Spectrom. 2007. PMID: 17200996
-
Evaluation of gas-phase rearrangement and competing fragmentation reactions on protein phosphorylation site assignment using collision induced dissociation-MS/MS and MS3.Anal Chem. 2008 Dec 15;80(24):9735-47. doi: 10.1021/ac801768s. Anal Chem. 2008. PMID: 19012417
-
Femtosecond laser-induced ionization/dissociation tandem mass spectrometry (fsLID-MS/MS) of deprotonated phosphopeptide anions.Rapid Commun Mass Spectrom. 2013 Dec 30;27(24):2807-17. doi: 10.1002/rcm.6750. Rapid Commun Mass Spectrom. 2013. PMID: 24214867
-
Phosphopeptide fragmentation and analysis by mass spectrometry.J Mass Spectrom. 2009 Jun;44(6):861-78. doi: 10.1002/jms.1599. J Mass Spectrom. 2009. PMID: 19504542 Review.
-
Ion/ion chemistry of high-mass multiply charged ions.Mass Spectrom Rev. 1998 Nov-Dec;17(6):369-407. doi: 10.1002/(SICI)1098-2787(1998)17:6<369::AID-MAS1>3.0.CO;2-J. Mass Spectrom Rev. 1998. PMID: 10360331 Review.
Cited by
-
Ultraviolet, Infrared, and High-Low Energy Photodissociation of Post-Translationally Modified Peptides.J Am Soc Mass Spectrom. 2018 Feb;29(2):270-283. doi: 10.1007/s13361-017-1794-9. Epub 2017 Oct 4. J Am Soc Mass Spectrom. 2018. PMID: 28980177
-
The Role of Electron Transfer Dissociation in Modern Proteomics.Anal Chem. 2018 Jan 2;90(1):40-64. doi: 10.1021/acs.analchem.7b04810. Epub 2017 Dec 12. Anal Chem. 2018. PMID: 29172454 Free PMC article. Review. No abstract available.
-
Characterization of Phosphorylated Peptides by Electron-Activated and Ultraviolet Dissociation Mass Spectrometry: A Comparative Study with Collision-Induced Dissociation.J Am Soc Mass Spectrom. 2024 May 1;35(5):1040-1054. doi: 10.1021/jasms.4c00048. Epub 2024 Apr 16. J Am Soc Mass Spectrom. 2024. PMID: 38626331 Free PMC article.
-
CE-MS/MS and CE-timsTOF to separate and characterize intramolecular disulfide bridges of monoclonal antibody subunits and their application for the assessment of subunit reduction protocols.Anal Bioanal Chem. 2024 Mar;416(7):1599-1612. doi: 10.1007/s00216-024-05161-8. Epub 2024 Feb 1. Anal Bioanal Chem. 2024. PMID: 38296860 Free PMC article.
-
Recent Developments in Gas-Phase Ion/Ion Reactions for Analytical Mass Spectrometry.Anal Chem. 2020 Jan 7;92(1):252-266. doi: 10.1021/acs.analchem.9b05014. Epub 2019 Nov 26. Anal Chem. 2020. PMID: 31693342 Free PMC article. Review.
References
-
- Cohen P. Nat Cell Biol. 2002;4:E127–E130. - PubMed
-
- Ubersax JA, Ferrell JE., Jr Nat Rev Mol Cell Biol. 2007;8:530–541. - PubMed
-
- Johnson LN. Biochem Soc Trans. 2009;37:627–641. - PubMed
-
- Hunter T. Cell. 1995;80:225–236. - PubMed
-
- Lahiry P, Torkamani A, Schork NJ, Hegele RA. Nat Rev Genet. 2010;11:60–74. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

