Intravenous pamidronate versus oral and intravenous clodronate in bone metastatic breast cancer: a randomized, open-label, non-inferiority Phase III trial
- PMID: 27468239
- PMCID: PMC4944913
- DOI: 10.2147/OTT.S103130
Intravenous pamidronate versus oral and intravenous clodronate in bone metastatic breast cancer: a randomized, open-label, non-inferiority Phase III trial
Abstract
Purpose: Patients with metastasized breast cancer often suffer from discomfort caused by metastatic bone disease. Thus, osteoprotection is an important part of therapy in breast cancer metastasized to bone, and bisphosphonates (BPs) are a major therapeutic option. In this study, our objectives were to compare the side effects of oral versus intravenous BP treatment and to assess their clinical effectiveness.
Patients and methods: In this prospective randomized, open-label, non-inferiority trial, we enrolled breast cancer patients with at least one bone metastasis and an Eastern Cooperative Oncology Group performance status of 0-2. Patients were randomly assigned to one of the three treatment groups: A, 60 mg pamidronate intravenously q3w; B-iv, 900 mg clodronate intravenously q3w; and B-o, 2,400 mg oral clodronate daily. Assessments were performed at baseline and every 3 months thereafter.
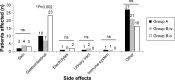
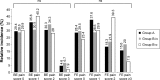
Results: Between 1995 and 1999, 321 patients with confirmed bone metastases from breast cancer were included in the study. At first follow-up, gastrointestinal (GI) tract side effects were most common, and adverse effects on the GI tract were more frequent in the oral treatment group (P=0.002 and P<0.001, respectively). There were no statistically significant differences among the treatment cohorts for other documented side effects (skin, serum electrolytes, urinary tract, immune system, and others). No significant differences in clinical effectiveness of BP treatment, as assessed by pain score, were detected among the groups; however, pathologic fractures were more effectively prevented by intravenous than oral BP administration (P=0.03). Noncompliance rates were similar among the study cohorts.
Conclusion: We conclude that oral BP treatment is significantly associated with higher rates of adverse GI side effects. Additionally, our data indicate that intravenous BP administration is more effective than oral treatment in prevention of pathologic fractures; hence, oral administration should be considered with caution.
Keywords: adverse effects; bisphosphonates; clinical effectiveness; metastatic bone disease.
Figures
References
-
- Robert Koch Institute Breast cancer. 2015. [Accessed May 20, 2015]. Available from: http://www.krebsdaten.de/Krebs/EN/Content/Cancer_sites/Breast_cancer/bre....
-
- Psaila B, Kaplan RN, Port ER, Lyden D. Priming the ‘soil’ for breast cancer metastasis: the pre-metastatic niche. Breast Dis. 2006;26:65–74. - PubMed
-
- Coleman RE. Skeletal complications of malignancy. Cancer. 1997;80(8 Suppl):1588–1594. - PubMed
-
- Plunkett TA, Smith P, Rubens RD. Risk of complications from bone metastases in breast cancer. Implications for management. Eur J Cancer. 2000;36(4):476–482. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

