Silica Coating of Nonsilicate Nanoparticles for Resin-Based Composite Materials
- PMID: 27470069
- PMCID: PMC5076750
- DOI: 10.1177/0022034516662022
Silica Coating of Nonsilicate Nanoparticles for Resin-Based Composite Materials
Abstract
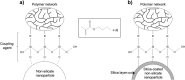
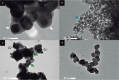
This study was designed to develop and characterize a silica-coating method for crystalline nonsilicate ceramic nanoparticles (Al2O3, TiO2, and ZrO2). The hypothesis was that the coated nonsilicate nanoparticles would stably reinforce a polymeric matrix due to effective silanation. Silica coating was applied via a sol-gel method, with tetraethyl orthosilicate as a silica precursor, followed by heat treatment. The chemical and microstructural characteristics of the nanopowders were evaluated before and after silica coating through x-ray diffraction, BET (Brunauer-Emmett-Teller), energy-dispersive x-ray spectroscopy, field emission scanning electron microscopy, and transmission electron microscopy analyses. Coated and noncoated nanoparticles were silanated before preparation of hybrid composites, which contained glass microparticles in addition to the nanoparticles. The composites were mechanically tested in 4-point bending mode after aging (10,000 thermal cycles). Results of all chemical and microstructural analyses confirmed the successful obtaining of silica-coated nanoparticles. Two distinct aspects were observed depending on the type of nanoparticle tested: 1) formation of a silica shell on the surface of the particles and 2) nanoparticle clusters embedded into a silica matrix. The aged hybrid composites formulated with the coated nanoparticles showed improved flexural strength (10% to 30% higher) and work of fracture (35% to 40% higher) as compared with composites formulated with noncoated nanoparticles. The tested hypothesis was confirmed: silanated silica-coated nonsilicate nanoparticles yielded stable reinforcement of dimethacrylate polymeric matrix due to effective silanation. The silica-coating method presented here is a versatile and promising novel strategy for the use of crystalline nonsilicate ceramics as a reinforcing phase of polymeric composite biomaterials.
Keywords: CAD-CAM; microscopy; nanotechnology; polymers; silane; surface properties.
Conflict of interest statement
The authors declare no potential conflicts of interest with respect to the authorship and/or publication of this article.
Figures

Similar articles
-
Effect of surface treatments on the shear bond strength of luting cements to Y-TZP ceramic.J Prosthet Dent. 2015 Mar;113(3):212-9. doi: 10.1016/j.prosdent.2014.09.012. Epub 2014 Nov 18. J Prosthet Dent. 2015. PMID: 25453565
-
Effect of zirconium-oxide ceramic surface treatments on the bond strength to adhesive resin.J Prosthet Dent. 2006 Jun;95(6):430-6. doi: 10.1016/j.prosdent.2006.03.016. J Prosthet Dent. 2006. PMID: 16765155
-
Mechanical properties of dental resin/composite containing urchin-like hydroxyapatite.Dent Mater. 2014 Dec;30(12):1358-68. doi: 10.1016/j.dental.2014.10.003. Dent Mater. 2014. PMID: 25458352
-
Bio-templated silica composites for next-generation biomedical applications.Adv Colloid Interface Sci. 2017 Nov;249:272-289. doi: 10.1016/j.cis.2017.04.011. Epub 2017 Apr 29. Adv Colloid Interface Sci. 2017. PMID: 28499603 Review.
-
Preparation and biomedical applications of core-shell silica/magnetic nanoparticle composites.J Nanosci Nanotechnol. 2012 Apr;12(4):2964-72. doi: 10.1166/jnn.2012.6428. J Nanosci Nanotechnol. 2012. PMID: 22849053 Review.
Cited by
-
The science of printing and polishing 3D printed dentures.F1000Res. 2025 Jan 6;13:1266. doi: 10.12688/f1000research.157596.2. eCollection 2024. F1000Res. 2025. PMID: 39839733 Free PMC article. Review.
-
Non-silicate nanoparticles for improved nanohybrid resin composites.Dent Mater. 2020 Oct;36(10):1314-1321. doi: 10.1016/j.dental.2020.07.001. Epub 2020 Aug 3. Dent Mater. 2020. PMID: 32758374 Free PMC article.
-
Sol-Gel Synthesis and Characterization of YSZ Nanofillers for Dental Cements at Different Temperatures.Dent J (Basel). 2021 Oct 29;9(11):128. doi: 10.3390/dj9110128. Dent J (Basel). 2021. PMID: 34821592 Free PMC article.
-
Physical and structural characterization of bis-acryl composite resin.Sci Rep. 2024 Apr 6;14(1):8075. doi: 10.1038/s41598-024-58649-9. Sci Rep. 2024. PMID: 38580685 Free PMC article.
-
Biocompatible Nanocapsules for Self-Healing Dental Resins and Bone Cements.ACS Omega. 2022 Aug 31;7(36):31726-31735. doi: 10.1021/acsomega.2c02080. eCollection 2022 Sep 13. ACS Omega. 2022. PMID: 36120077 Free PMC article.
References
-
- Abboud M, Turner M, Duguet E, Fontanille M. 1997. PMMA-based composite materials with reactive ceramic fillers: 1. Chemical modification and characterisation of ceramic particles. J Mater Chem. 7(8):1527–1532.
-
- Broutman LJ, Agarwal BD. 1974. Theoretical study of effect of an interfacial layer on properties of composites. Polym Eng Sci. 14(8):581–588.
-
- Brown SK. 1980. Mechanisms of fracture in filled thermosetting resins. Brit Polym J. 12(1):24–30.
-
- Cava S, Tebcherani SM, Souza IA, Pianaro SA, Paskocimas CA, Longo E, Varela JA. 2007. Structural characterization of phase transition of Al2O3 nanopowders obtained by polymeric precursor method. Mater Chem Phys. 103(2–3):394–399.
-
- Coldea A, Fischer J, Swain MV, Thiel N. 2015. Damage tolerance of indirect restorative materials (including PICN) after simulated bur adjustments. Dent Mater. 31(6):684–694. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

