Formation of polarity convergences underlying shoot outgrowths
- PMID: 27478985
- PMCID: PMC4969039
- DOI: 10.7554/eLife.18165
Formation of polarity convergences underlying shoot outgrowths
Abstract
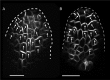
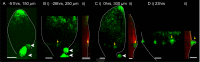
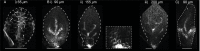
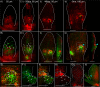
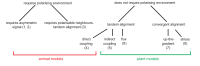
The development of outgrowths from plant shoots depends on formation of epidermal sites of cell polarity convergence with high intracellular auxin at their centre. A parsimonious model for generation of convergence sites is that cell polarity for the auxin transporter PIN1 orients up auxin gradients, as this spontaneously generates convergent alignments. Here we test predictions of this and other models for the patterns of auxin biosynthesis and import. Live imaging of outgrowths from kanadi1 kanadi2 Arabidopsis mutant leaves shows that they arise by formation of PIN1 convergence sites within a proximodistal polarity field. PIN1 polarities are oriented away from regions of high auxin biosynthesis enzyme expression, and towards regions of high auxin importer expression. Both expression patterns are required for normal outgrowth emergence, and may form part of a common module underlying shoot outgrowths. These findings are more consistent with models that spontaneously generate tandem rather than convergent alignments.
Keywords: A. thaliana; CUC; auxin; developmental biology; leaf development; plant biology; polarity; stem cells.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures





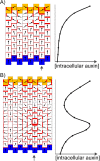

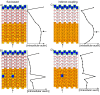
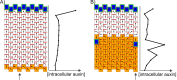
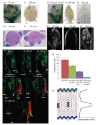

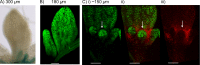


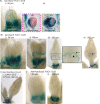
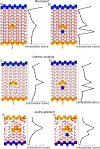
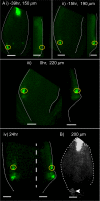
References
Publication types
MeSH terms
Substances
Grants and funding
- BBS/E/J/00000152/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- BBS/E/J/000C0645/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- BB/L008920/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- BB/F005997/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

