Pegylated-asparaginase during induction therapy for adult acute lymphoblastic leukaemia: toxicity data from the UKALL14 trial
- PMID: 27480385
- PMCID: PMC5154375
- DOI: 10.1038/leu.2016.219
Pegylated-asparaginase during induction therapy for adult acute lymphoblastic leukaemia: toxicity data from the UKALL14 trial
Abstract
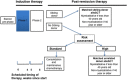
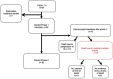
Safety and efficacy data on pegylated asparaginase (PEG-ASP) in adult acute lymphoblastic leukaemia (ALL) induction regimens are limited. The UK National Cancer Research Institute UKALL14 trial NCT01085617 prospectively evaluated the tolerability of 1000 IU/m2 PEG-ASP administered on days 4 and 18 as part of a five-drug induction regimen in adults aged 25-65 years with de novo ALL. Median age was 46.5 years. Sixteen of the 90 patients (median age 56 years) suffered treatment-related mortality during initial induction therapy. Eight of the 16 died of sepsis in combination with hepatotoxicity. Age and Philadelphia (Ph) status were independent variables predicting induction death >40 versus ⩽40 years, odds ratio (OR) 18.5 (2.02-169.0), P=0.01; Ph- versus Ph+ disease, OR 13.60 (3.52-52.36), P<0.001. Of the 74 patients who did not die, 37 (50.0%) experienced at least one grade 3/4 PEG-ASP-related adverse event, most commonly hepatotoxicity (36.5%, n=27). A single dose of PEG-ASP achieved trough therapeutic enzyme levels in 42/49 (86%) of the patients tested. Although PEG-ASP delivered prolonged asparaginase activity in adults, it was difficult to administer safely as part of the UKALL14 intensive multiagent regimen to those aged >40 years. It proved extremely toxic in patients with Ph+ ALL, possibly owing to interaction with imatinib.
Conflict of interest statement
Fielding received lab funding from Medac GmBH to carry out part of this work and that company were the supplier of PEG-ASP at the time of this work. The remaining authors declare no conflict of interest.
Figures
References
-
- Sallan SE, Hitchcock-Bryan S, Gelber R, Cassady JR, Frei E 3rd, Nathan DG. Influence of intensive asparaginase in the treatment of childhood non-T-cell acute lymphoblastic leukemia. Cancer Res 1983; 43: 5601–5607. - PubMed
-
- Avramis VI, Sencer S, Periclou AP, Sather H, Bostrom BC, Cohen LJ et al. A randomized comparison of native Escherichia coli asparaginase and polyethylene glycol conjugated asparaginase for treatment of children with newly diagnosed standard-risk acute lymphoblastic leukemia: a Children's Cancer Group study. Blood 2002; 99: 1986–1994. - PubMed
-
- Seibel NL. Treatment of acute lymphoblastic leukemia in children and adolescents: peaks and pitfalls. Hematology Am Soc Hematol Educ Program 2008, 374–380. - PubMed
-
- Avramis VI, Spence SA. Clinical pharmacology of asparaginases in the United States: asparaginase population pharmacokinetic and pharmacodynamic (PK-PD) models (NONMEM) in adult and pediatric ALL patients. J Pediatr Hematol Oncol 2007; 29: 239–247. - PubMed
-
- Kurtzberg J, Asselin B, Bernstein M, Buchanan GR, Pollock BH, Camitta BM. Polyethylene glycol-conjugated L-asparaginase versus native L-asparaginase in combination with standard agents for children with acute lymphoblastic leukemia in second bone marrow relapse: a Children's Oncology Group Study (POG 8866). J Pediatr Hematol Oncol 2011; 33: 610–616. - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

