Phase II Study of Alemtuzumab (CAMPATH-1) in Patients with HTLV-1-Associated Adult T-cell Leukemia/lymphoma
- PMID: 27486175
- PMCID: PMC5215752
- DOI: 10.1158/1078-0432.CCR-16-1022
Phase II Study of Alemtuzumab (CAMPATH-1) in Patients with HTLV-1-Associated Adult T-cell Leukemia/lymphoma
Abstract
Purpose: Therapeutic regimens for adult T-cell leukemia/lymphoma (ATL) are limited with unsatisfactory results, thereby warranting development of novel therapies. This study investigated antitumor activity and toxicity of alemtuzumab with regard to response, duration of response, progression-free survival, and overall survival in patients with human T-cell lymphotropic virus-1 (HTLV-1)-associated ATL.
Experimental design: Twenty-nine patients with chronic, acute, and lymphomatous types of ATL were enrolled in a single-institution, nonrandomized, open-label phase II trial wherein patients received intravenous alemtuzumab 30 mg three times weekly for a maximum of 12 weeks.
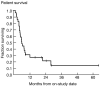
Results: Twenty-nine patients were evaluable for response and toxicity. The overall objective response was 15 of 29 patients [95% confidence interval (CI), 32.5%-70.6%]. The 15 patients who responded manifested a median time to response of 1.1 months. Median response duration was 1.4 months for the whole group and 14.5 months among responders. Median progression-free survival was 2.0 months. Median overall survival was 5.9 months. The most common adverse events were 2 with vasovagal episodes (7%) and 3 with hypotensive episodes (10%), leukopenia (41%) grade 3 and (17%) grade 4, lymphocytopenia (59%) grade 3, neutropenia (31%) grade 3, anemia (24%), and thrombocytopenia (10%). All patients developed cytomegalovirus antigenemia (CMV). Three were symptomatic and all responded to antiviral therapy. Grade 3 or 4 infections were reported in 4 (14%) of patients.
Conclusions: Alemtuzumab induced responses in patients with acute HTLV-1-associated ATL with acceptable toxicity, but with short duration of responses. These studies support inclusion of alemtuzumab in novel multidrug therapies for ATL. Clin Cancer Res; 23(1); 35-42. ©2016 AACR.
©2016 American Association for Cancer Research.
Figures
Similar articles
-
Radiation therapy for the management of patients with HTLV-1-associated adult T-cell leukemia/lymphoma.Blood. 2012 Aug 30;120(9):1816-9. doi: 10.1182/blood-2012-01-401349. Epub 2012 Jun 22. Blood. 2012. PMID: 22730536 Free PMC article.
-
Epidemiology, clinical features, and outcome of HTLV-1-related ATLL in an area of prevalence in the United States.Blood Adv. 2018 Mar 27;2(6):607-620. doi: 10.1182/bloodadvances.2017011106. Blood Adv. 2018. PMID: 29545256 Free PMC article.
-
Clinical significance of CD70 expression on T cells in human T-lymphotropic virus type-1 carriers and adult T cell leukemia/ lymphoma patients.Leuk Lymphoma. 2016;57(3):685-91. doi: 10.3109/10428194.2015.1063140. Epub 2015 Jul 25. Leuk Lymphoma. 2016. PMID: 26077361
-
Human T-cell leukaemia virus type I and adult T-cell leukaemia-lymphoma.Lancet Oncol. 2014 Oct;15(11):e517-26. doi: 10.1016/S1470-2045(14)70202-5. Lancet Oncol. 2014. PMID: 25281470 Review.
-
Overview of Targeted Therapies for Adult T-Cell Leukemia/Lymphoma.Methods Mol Biol. 2017;1582:197-216. doi: 10.1007/978-1-4939-6872-5_15. Methods Mol Biol. 2017. PMID: 28357672 Review.
Cited by
-
Novel Treatments of Adult T Cell Leukemia Lymphoma.Front Microbiol. 2020 May 28;11:1062. doi: 10.3389/fmicb.2020.01062. eCollection 2020. Front Microbiol. 2020. PMID: 32547515 Free PMC article. Review.
-
Interplay between innate immunity and the viral oncoproteins Tax and HBZ in the pathogenesis and therapeutic response of HTLV-1 associated adult T cell leukemia.Front Immunol. 2022 Jul 22;13:957535. doi: 10.3389/fimmu.2022.957535. eCollection 2022. Front Immunol. 2022. PMID: 35935975 Free PMC article. Review.
-
Future Perspectives on Drug Targeting in Adult T Cell Leukemia-Lymphoma.Front Microbiol. 2018 May 9;9:925. doi: 10.3389/fmicb.2018.00925. eCollection 2018. Front Microbiol. 2018. PMID: 29867836 Free PMC article. Review.
-
Antibody-Based Therapies for Peripheral T-Cell Lymphoma.Cancers (Basel). 2024 Oct 15;16(20):3489. doi: 10.3390/cancers16203489. Cancers (Basel). 2024. PMID: 39456582 Free PMC article. Review.
-
One step further in targeting acute leukemia by combining antibody-based immunotherapies and small molecule inhibitors.Cancer Cell Int. 2025 Jul 7;25(1):254. doi: 10.1186/s12935-025-03869-w. Cancer Cell Int. 2025. PMID: 40624659 Free PMC article. Review.
References
-
- Uchiyama T, Yodoi J, Sagawa K, Takatsuki K, Uchino H. Adult T-cell leukemia: clinical and hematologic features of 16 cases. Blood. 1977;50(3):481–492. - PubMed
-
- Yamada Y, Tomonaga M, Fukuda H, Hanada S, Utsunomiya A, Tara M, et al. A new G-CSF-supported combination therapy, LSG15, for adult T-cell leukaemia-lymphoma: Japan Clinical Oncology Group Study 9303. Br J Haematol. 2001;113(2):375–382. - PubMed
-
- Broder S, Bunn PA, Jr, Jaffe ES, Blattner W, Gallo RC, Wong-Staal F, et al. NIH conference T-cell lymphoproliferative syndrome associated with human T-cell leukemia/lymphoma virus. Ann Intern Med. 1984;100:543–557. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources