Obesity Outweighs Protection Conferred by Adjuvanted Influenza Vaccination
- PMID: 27486196
- PMCID: PMC4981723
- DOI: 10.1128/mBio.01144-16
Obesity Outweighs Protection Conferred by Adjuvanted Influenza Vaccination
Abstract
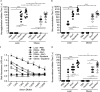
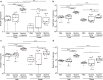
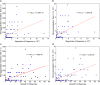
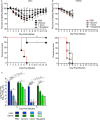
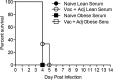
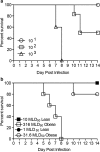
Obesity is a risk factor for developing severe influenza virus infection, making vaccination of utmost importance for this high-risk population. However, vaccinated obese animals and adults have decreased neutralizing antibody responses. In these studies, we tested the hypothesis that the addition of either alum or a squalene-based adjuvant (AS03) to an influenza vaccine would improve neutralizing antibody responses and protect obese mice from challenge. Our studies demonstrate that adjuvanted vaccine does increase both neutralizing and nonneutralizing antibody levels compared to vaccine alone. Although obese mice mount significantly decreased virus-specific antibody responses, both the breadth and the magnitude of the responses against hemagglutinin (HA) and neuraminidase (NA) are decreased compared to the responses in lean mice. Importantly, even with a greater than fourfold increase in neutralizing antibody levels, obese mice are not protected against influenza virus challenge and viral loads remain elevated in the respiratory tract. Increasing the antigen dose affords no added protection, and a decreasing viral dose did not fully mitigate the increased mortality seen in obese mice. Overall, these studies highlight that, while the use of an adjuvant does improve seroconversion, vaccination does not fully protect obese mice from influenza virus challenge, possibly due to the increased sensitivity of obese animals to infection. Given the continued increase in the global obesity epidemic, our findings have important implications for public health.
Importance: Vaccination is the most effective strategy for preventing influenza virus infection and is a key component for pandemic preparedness. However, vaccines may fail to provide optimal protection in high-risk groups, including overweight and obese individuals. Given the worldwide obesity epidemic, it is imperative that we understand and improve vaccine efficacy. No work to date has investigated whether adjuvants increase the protective capacity of influenza vaccines in the obese host. In these studies, we show that adjuvants increased the neutralizing and nonneutralizing antibody responses during vaccination of lean and obese mice to levels considered "protective," and yet, obese mice still succumbed to infection. This vulnerability is likely due to a combination of factors, including the increased susceptibility of obese animals to develop severe and even lethal disease when infected with very low viral titers. Our studies highlight the critical public health need to translate these findings and better understand vaccination in this increasing population.
Copyright © 2016 Karlsson et al.
Figures



Similar articles
-
Squalene-containing licensed adjuvants enhance strain-specific antibody responses against the influenza hemagglutinin and induce subtype-specific antibodies against the neuraminidase.Vaccine. 2016 Oct 17;34(44):5329-5335. doi: 10.1016/j.vaccine.2016.08.082. Epub 2016 Sep 8. Vaccine. 2016. PMID: 27616472
-
Comparison of AS03 and Alum on immune responses elicited by A/H3N2 split influenza vaccine in young, mature and aged BALB/c mice.Vaccine. 2016 Mar 14;34(12):1444-51. doi: 10.1016/j.vaccine.2016.02.012. Epub 2016 Feb 9. Vaccine. 2016. PMID: 26873056
-
Immunogenicity and Safety of an AS03-Adjuvanted H7N9 Pandemic Influenza Vaccine in a Randomized Trial in Healthy Adults.J Infect Dis. 2016 Dec 1;214(11):1717-1727. doi: 10.1093/infdis/jiw414. Epub 2016 Sep 7. J Infect Dis. 2016. PMID: 27609809 Free PMC article. Clinical Trial.
-
An indirect comparison meta-analysis of AS03 and MF59 adjuvants in pandemic influenza A(H1N1)pdm09 vaccines.Vaccine. 2019 Jul 18;37(31):4246-4255. doi: 10.1016/j.vaccine.2019.06.039. Epub 2019 Jun 26. Vaccine. 2019. PMID: 31253447 Review.
-
Narcolepsy, 2009 A(H1N1) pandemic influenza, and pandemic influenza vaccinations: what is known and unknown about the neurological disorder, the role for autoimmunity, and vaccine adjuvants.J Autoimmun. 2014 May;50:1-11. doi: 10.1016/j.jaut.2014.01.033. Epub 2014 Feb 19. J Autoimmun. 2014. PMID: 24559657 Review.
Cited by
-
Effect of pioglitazone on inflammatory response and clinical outcome in T2DM patients with COVID-19: a randomized multicenter double-blind clinical trial.Front Immunol. 2024 Sep 6;15:1369918. doi: 10.3389/fimmu.2024.1369918. eCollection 2024. Front Immunol. 2024. PMID: 39308871 Free PMC article. Clinical Trial.
-
Host nutritional status affects alphavirus virulence, transmission, and evolution.PLoS Pathog. 2019 Nov 11;15(11):e1008089. doi: 10.1371/journal.ppat.1008089. eCollection 2019 Nov. PLoS Pathog. 2019. PMID: 31710653 Free PMC article.
-
COVID-19 infection, progression, and vaccination: Focus on obesity and related metabolic disturbances.Obes Rev. 2021 Oct;22(10):e13313. doi: 10.1111/obr.13313. Epub 2021 Jul 16. Obes Rev. 2021. PMID: 34269511 Free PMC article. Review.
-
Obesity Is Associated with an Impaired Baseline Repertoire of Anti-Influenza Virus Antibodies.Microbiol Spectr. 2023 Jun 15;11(3):e0001023. doi: 10.1128/spectrum.00010-23. Epub 2023 Apr 26. Microbiol Spectr. 2023. PMID: 37098954 Free PMC article.
-
Diet-induced obesity affects influenza disease severity and transmission dynamics in ferrets.Sci Adv. 2024 May 10;10(19):eadk9137. doi: 10.1126/sciadv.adk9137. Epub 2024 May 10. Sci Adv. 2024. PMID: 38728395 Free PMC article.
References
-
- Morgan OW, Bramley A, Fowlkes A, Freedman DS, Taylor TH, Gargiullo P, Belay B, Jain S, Cox C, Kamimoto L, Fiore A, Finelli L, Olsen SJ, Fry AM. 2010. Morbid obesity as a risk factor for hospitalization and death due to 2009 pandemic influenza A(H1N1) disease. PLoS One 5:e9694. doi:10.1371/journal.pone.0009694. - DOI - PMC - PubMed
-
- Louie JK, Acosta M, Winter K, Jean C, Gavali S, Schechter R, Vugia D, Harriman K, Matyas B, Glaser CA, Samuel MC, Rosenberg J, Talarico J, Hatch D, California Pandemic (H1N1) Working Group . 2009. Factors associated with death or hospitalization due to pandemic 2009 influenza A(H1N1) infection in California. JAMA 302:1896–1902. doi:10.1001/jama.2009.1583. - DOI - PubMed
-
- Liu B, Havers F, Chen E, Yuan Z, Yuan H, Ou J, Shang M, Kang K, Liao K, Liu F, Li D, Ding H, Zhou L, Zhu W, Ding F, Zhang P, Wang X, Yao J, Xiang N, Zhou S, Liu X, Song Y, Su H, Wang R, Cai J, Cao Y, Wang X, Bai T, Wang J, Feng Z, Zhang Y, Widdowson M-A, Li Q. 2014. Risk factors for influenza A(H7N9) disease—China, 2013. Clin Infect Dis 59:787–794. doi:10.1093/cid/ciu423. - DOI - PubMed
-
- World Health Organization 2015. Obesity and overweight. Fact sheet 311. WHO, Geneva, Switzerland: Accessed 20 February 2016 http://www.who.int/mediacentre/factsheets/fs311/en/. Accessed 20 February 2015.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

