Skeletal complications in cancer patients with bone metastases
- PMID: 27488133
- PMCID: PMC5718365
- DOI: 10.1111/iju.13170
Skeletal complications in cancer patients with bone metastases
Abstract
As a result of significant improvements in current therapies, the life expectancy of cancer patients with bone metastases has dramatically improved. Unfortunately, these patients often experience skeletal complications that significantly impair their quality of life. The major skeletal complications associated with bone metastases include: cancer-induced bone pain, hypercalcemia, pathological bone fractures, metastatic epidural spinal cord compression and cancer cachexia. Once cancer cells invade the bone, they perturb the normal physiology of the marrow microenvironment, resulting in bone destruction, which is believed to be a direct cause of skeletal complications. However, full understanding of the mechanisms responsible for these complications remains unknown. In the present review, we discuss the complications associated with bone metastases along with matched conventional therapeutic strategies. A better understanding of this topic is crucial, as targeting skeletal complications can improve both the morbidity and mortality of patients suffering from bone metastases.
Keywords: bone metastasis; cancer-induced bone pain; hypercalcemia; metastatic epidural spinal cord compression; pathological bone fractures; skeletal complications.
© 2016 The Japanese Urological Association.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures
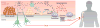
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

