LTβR controls thymic portal endothelial cells for haematopoietic progenitor cell homing and T-cell regeneration
- PMID: 27493002
- PMCID: PMC4980457
- DOI: 10.1038/ncomms12369
LTβR controls thymic portal endothelial cells for haematopoietic progenitor cell homing and T-cell regeneration
Abstract
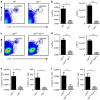
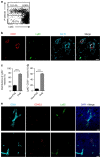
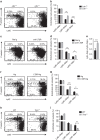
Continuous thymic homing of haematopoietic progenitor cells (HPCs) via the blood is critical for normal T-cell development. However, the nature and the differentiation programme of specialized thymic endothelial cells (ECs) controlling this process remain poorly understood. Here using conditional gene-deficient mice, we find that lymphotoxin beta receptor (LTβR) directly controls thymic ECs to guide HPC homing. Interestingly, T-cell deficiency or conditional ablation of T-cell-engaged LTβR signalling results in a defect in thymic HPC homing, suggesting the feedback regulation of thymic progenitor homing by thymic products. Furthermore, we identify and characterize a special thymic portal EC population with features that guide HPC homing. LTβR is essential for the differentiation and homeostasis of these thymic portal ECs. Finally, we show that LTβR is required for T-cell regeneration on irradiation-induced thymic injury. Together, these results uncover a cellular and molecular pathway that governs thymic EC differentiation for HPC homing.
Figures



References
-
- Martins V. C. et al. Cell competition is a tumour suppressor mechanism in the thymus. Nature 509, 465–470 (2014). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

