The organizing actions of adolescent gonadal steroid hormones on brain and behavioral development
- PMID: 27497718
- PMCID: PMC5074860
- DOI: 10.1016/j.neubiorev.2016.07.036
The organizing actions of adolescent gonadal steroid hormones on brain and behavioral development
Abstract
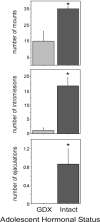
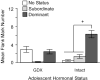
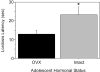
Adolescence is a developmental period characterized by dramatic changes in cognition, risk-taking and social behavior. Although gonadal steroid hormones are well-known mediators of these behaviors in adulthood, the role gonadal steroid hormones play in shaping the adolescent brain and behavioral development has only come to light in recent years. Here we discuss the sex-specific impact of gonadal steroid hormones on the developing adolescent brain. Indeed, the effects of gonadal steroid hormones during adolescence on brain structure and behavioral outcomes differs markedly between the sexes. Research findings suggest that adolescence, like the perinatal period, is a sensitive period for the sex-specific effects of gonadal steroid hormones on brain and behavioral development. Furthermore, evidence from studies on male sexual behavior suggests that adolescence is part of a protracted postnatal sensitive period that begins perinatally and ends following adolescence. As such, the perinatal and peripubertal periods of brain and behavioral organization likely do not represent two discrete sensitive periods, but instead are the consequence of normative developmental timing of gonadal hormone secretions in males and females.
Keywords: Activational-organizational hypothesis; Adolescence; Agonistic behavior; Anxiety-like behavior; Cortex; Estrogen; Ingestive behavior; Sensitive periods; Sexual behavior; Synaptic pruning; Testosterone.
Copyright © 2016 Elsevier Ltd. All rights reserved.
Figures

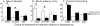

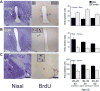
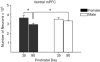
References
-
- Adkins-Regan E, Orgeur P, Signoret JP. Sexual differentiation of reproductive behavior in pigs: defeminizing effects of prepubertal estradiol. Horm Behav. 1989;23:290–303. - PubMed
-
- Andersen SL. Trajectories of brain development: point of vulnerability or window of opportunity? Neurosci Biobehav Rev. 2003;27:3–18. - PubMed
-
- Andersen SL, Rutstein M, Benzo JM, Hostetter JC, Teicher MH. Sex differences in dopamine receptor overproduction and elimination. Neuroreport. 1997;8:1495–1498. - PubMed
-
- Andrews WW, Mizejewski GJ, Ojeda SR. Development of estradiol-positive feedback on luteinizing hormone release in the female rat: a quantitative study. Endocrinology. 1981;109:1404–1413. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

