The structural basis of the dominant negative phenotype of the Gαi1β1γ2 G203A/A326S heterotrimer
- PMID: 27498775
- PMCID: PMC5022103
- DOI: 10.1038/aps.2016.69
The structural basis of the dominant negative phenotype of the Gαi1β1γ2 G203A/A326S heterotrimer
Abstract
Aim: Dominant negative mutant G proteins have provided critical insight into the mechanisms of G protein-coupled receptor (GPCR) signaling, but the mechanisms underlying the dominant negative characteristics are not completely understood. The aim of this study was to determine the structure of the dominant negative Gαi1β1γ2 G203A/A326S complex (Gi-DN) and to reveal the structural basis of the mutation-induced phenotype of Gαi1β1γ2.
Methods: The three subunits of the Gi-DN complex were co-expressed with a baculovirus expression system. The Gi-DN heterotrimer was purified, and the structure of its complex with GDP was determined through X-ray crystallography.
Results: The Gi-DN heterotrimer structure revealed a dual mechanism underlying the dominant negative characteristics. The mutations weakened the hydrogen bonding network between GDP/GTP and the binding pocket residues, and increased the interactions in the Gα-Gβγ interface. Concomitantly, the Gi-DN heterotrimer adopted a conformation, in which the C-terminus of Gαi and the N-termini of both the Gβ and Gγ subunits were more similar to the GPCR-bound state compared with the wild type complex. From these structural observations, two additional mutations (T48F and D272F) were designed that completely abolish the GDP binding of the Gi-DN heterotrimer.
Conclusion: Overall, the results suggest that the mutations impede guanine nucleotide binding and Gα-Gβγ protein dissociation and favor the formation of the G protein/GPCR complex, thus blocking signal propagation. In addition, the structure provides a rationale for the design of other mutations that cause dominant negative effects in the G protein, as exemplified by the T48F and D272F mutations.
Figures


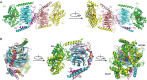





References
-
- Sunahara RK, Tesmer JJ, Gilman AG, Sprang SR. Crystal structure of the adenylyl cyclase activator Gsalpha. Science 1997; 278: 1943–7. - PubMed
-
- Coleman DE, Berghuis AM, Lee E, Linder ME, Gilman AG, Sprang SR. Structures of active conformations of Gi alpha 1 and the mechanism of GTP hydrolysis. Science 1994; 265: 1405–12. - PubMed
-
- Noel JP, Hamm HE, Sigler PB. The 2.2 A crystal structure of transducin-alpha complexed with GTP gamma S. Nature 1993; 366: 654–63. - PubMed
-
- Mixon MB, Lee E, Coleman DE, Berghuis AM, Gilman AG, Sprang SR. Tertiary and quaternary structural changes in Gi alpha 1 induced by GTP hydrolysis. Science 1995; 270: 954–60. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

