Transcriptomic and Phenotypic Analysis Reveals New Functions for the Tat Pathway in Yersinia pseudotuberculosis
- PMID: 27501981
- PMCID: PMC5038016
- DOI: 10.1128/JB.00352-16
Transcriptomic and Phenotypic Analysis Reveals New Functions for the Tat Pathway in Yersinia pseudotuberculosis
Abstract
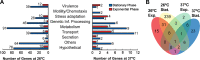
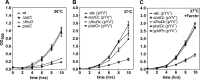
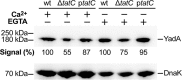
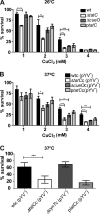
The twin-arginine translocation (Tat) system mediates the secretion of folded proteins that are identified via an N-terminal signal peptide in bacteria, plants, and archaea. Tat systems are associated with virulence in many bacterial pathogens, and our previous studies revealed that Tat-deficient Yersinia pseudotuberculosis was severely attenuated for virulence. Aiming to identify Tat-dependent pathways and phenotypes of relevance for in vivo infection, we analyzed the global transcriptome of parental and ΔtatC mutant strains of Y. pseudotuberculosis during exponential and stationary growth at 26°C and 37°C. The most significant changes in the transcriptome of the ΔtatC mutant were seen at 26°C during stationary-phase growth, and these included the altered expression of genes related to virulence, stress responses, and metabolism. Subsequent phenotypic analysis based on these transcriptome changes revealed several novel Tat-dependent phenotypes, including decreased YadA expression, impaired growth under iron-limited and high-copper conditions, as well as acidic pH and SDS. Several functionally related Tat substrates were also verified to contribute to these phenotypes. Interestingly, the phenotypic defects observed in the Tat-deficient strain were generally more pronounced than those in mutants lacking the Tat substrate predicted to contribute to that specific function. Altogether, this provides new insight into the impact of Tat deficiency on in vivo fitness and survival/replication of Y. pseudotuberculosis during infection.
Importance: In addition to its established role in mediating the secretion of housekeeping enzymes, the Tat system has been recognized as being involved in infection. In some clinically relevant bacteria, such as Pseudomonas spp., several key virulence determinants can readily be identified among the Tat substrates. In enteropathogens, such as Yersinia spp., there are no obvious virulence determinants among the Tat substrates. Tat mutants show no growth defect in vitro but are highly attenuated in in vivo This makes Tat an attractive target for the development of novel antimicrobials. Therefore, it is important to establish the causes of the attenuation. Here, we show that the attenuation is likely due to synergistic effects of different Tat-dependent phenotypes that each contributes to lowered in vivo fitness.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures
Similar articles
-
The Tat Substrate SufI Is Critical for the Ability of Yersinia pseudotuberculosis To Cause Systemic Infection.Infect Immun. 2017 Mar 23;85(4):e00867-16. doi: 10.1128/IAI.00867-16. Print 2017 Apr. Infect Immun. 2017. PMID: 28115509 Free PMC article.
-
The twin arginine translocation system is essential for virulence of Yersinia pseudotuberculosis.Infect Immun. 2006 Mar;74(3):1768-76. doi: 10.1128/IAI.74.3.1768-1776.2006. Infect Immun. 2006. PMID: 16495550 Free PMC article.
-
Genome Scale Analysis Reveals IscR Directly and Indirectly Regulates Virulence Factor Genes in Pathogenic Yersinia.mBio. 2021 Jun 29;12(3):e0063321. doi: 10.1128/mBio.00633-21. Epub 2021 Jun 1. mBio. 2021. PMID: 34060331 Free PMC article.
-
Twin arginine translocation in Yersinia.Adv Exp Med Biol. 2007;603:258-67. doi: 10.1007/978-0-387-72124-8_23. Adv Exp Med Biol. 2007. PMID: 17966422 Review.
-
Yersinia adhesin A (YadA)--beauty & beast.Int J Med Microbiol. 2015 Feb;305(2):252-8. doi: 10.1016/j.ijmm.2014.12.008. Epub 2014 Dec 24. Int J Med Microbiol. 2015. PMID: 25604505 Review.
Cited by
-
Disruption of the NlpD lipoprotein of the plague pathogen Yersinia pestis affects iron acquisition and the activity of the twin-arginine translocation system.PLoS Negl Trop Dis. 2019 Jun 6;13(6):e0007449. doi: 10.1371/journal.pntd.0007449. eCollection 2019 Jun. PLoS Negl Trop Dis. 2019. PMID: 31170147 Free PMC article.
-
Two Rieske Fe/S Proteins and TAT System in Mesorhizobium loti MAFF303099: Differential Regulation and Roles on Nodulation.Front Plant Sci. 2018 Nov 20;9:1686. doi: 10.3389/fpls.2018.01686. eCollection 2018. Front Plant Sci. 2018. PMID: 30515183 Free PMC article.
-
The Tat Substrate SufI Is Critical for the Ability of Yersinia pseudotuberculosis To Cause Systemic Infection.Infect Immun. 2017 Mar 23;85(4):e00867-16. doi: 10.1128/IAI.00867-16. Print 2017 Apr. Infect Immun. 2017. PMID: 28115509 Free PMC article.
-
Citrobacter freundii fitness during bloodstream infection.Sci Rep. 2018 Aug 7;8(1):11792. doi: 10.1038/s41598-018-30196-0. Sci Rep. 2018. PMID: 30087402 Free PMC article.
-
The temperature-dependent expression of type II secretion system controls extracellular product secretion and virulence in mesophilic Aeromonas salmonida SRW-OG1.Front Cell Infect Microbiol. 2022 Aug 1;12:945000. doi: 10.3389/fcimb.2022.945000. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35979091 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

